Recombinant human FGF-2 (145 aa) protein (Qk025)
Recombinant human FGF-2 (145 aa) protein (Qk025)Recombinant human FGF-2 (145 aa) protein (Qk025)
Price range: £150.00 through £735.00
Human fibroblast growth factor 2 (FGF-2/bFGF) protein (145 aa) is our gold-standard and recommended FGF-2 / bFGF protein for induced pluripotent stem cell (iPSC) and embryonic stem cell (ESC) maintenance, and iPSC and mesenchymal stem cell (MSC) proliferation and differentiation.
Highly pure and bioactive 17 kDa FGF-2 protein animal origin-free (AOF) and carrier protein-free (CF) comprising the core structured region (145 aa) sufficient for full biological activity.
Qkine FGF-2 is also available as cell therapy grade with extended quality testing and documentation – Qk025-CTG
In stock
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
Price range: £150.00 through £735.00
Buy online with secure credit card or purchase order. For any questions, please email orders@qkine.com
Summary:
- High purity human FGF-2 / basic FGF, 145 aa form comprising residues 144-288 (Uniprot: P09038)
- 17 kDa
>98%, by SDS-PAGE quantitative densitometry
Expressed in E. coli
Animal origin-free (AOF) and carrier protein-free
Manufactured in our Cambridge, UK laboratories
Lyophilized from Tris, NaCl, CyS, mannitol
- Resuspend in sterile-filtered water at >50 µg/ml, add carrier protein if desired, prepare single use aliquots and store frozen at -20 °C (short-term) or -80 °C (long-term).
Featured applications:
Expansion of induced pluripotent, embryonic and mesenchymal stem cells
- Recombinant human BMP-2 protein (Qk007)
- Recombinant human activin A protein (Qk001)
- Recombinant human TGF-β1 PLUS™ protein (Qk010)
- Recombinant human EGF protein (Qk011)
- Recombinant human BMP-4 protein (Qk038)
- Recombinant human FGF-18 protein (Qk069)
- Recombinant human G-CSF protein (Qk074)
- Recombinant human TGF-β2 protein (Qk072)
- Recombinant human betacellulin (Qk123)
- Recombinant human follistatin-resistant activin A (FRACTA) protein (Qk035)

FGF-2 activity was determined using the Promega serum response element luciferase reporter assay (*) in transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of FGF-2 for 6 hours. Firefly luciferase activity is measured and normalized to the control Renilla luciferase activity. Data from Qk025 lot #204706. EC50 = 0.24 ng/ml (14 pM).
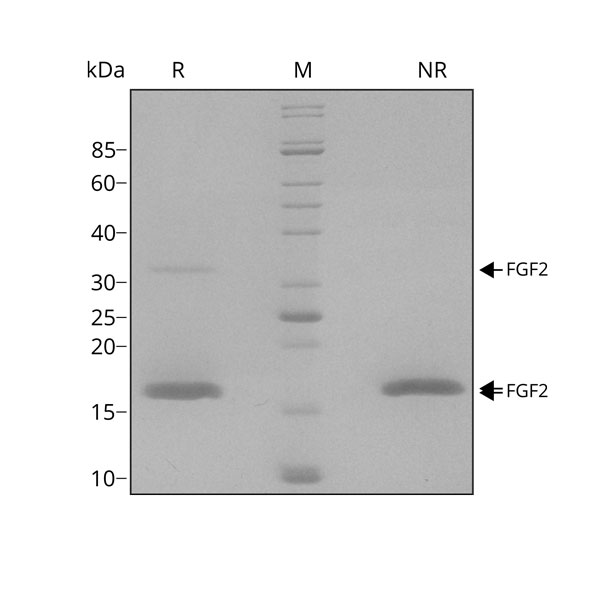
FGF-2 migrates as major band at 17 kDa in non-reducing (-βME) conditions and upon reduction (+βME). The higher molecular mass band at 35 kDa is a dimer that we always see in our highly purified human FGF-2, the presence of this does not affect biological activity. Purified recombinant protein (7 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptothanol, R) and non-reduced conditions (NR) and stained with Coomassie Brilliant Blue R250. Data from Qk025 lot #014
Further quality assays
Mass spectrometry: single species with expected mass
Recovery from stock vial: >95%
Endotoxin: <0.05 EU/μg protein
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.

Qkine human FGF-2 145 aa was found to be more bioactive than that from an alternative major supplier. Bioactivity was determined using the Promega serum response element luciferase reporter assay in transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of Qkine FGF-2 (Qk025, green) or alternative supplier FGF-2 (Supplier B, black) for 3 hours. Firefly luciferase activity was measured and normalized to control Renilla luciferase activity. Data from Qk025 lot #204697.
Protein background
Fibroblast growth factors (FGFs) are a family of growth factors which regulate a wide range of essential biological functions including cell proliferation and survival, migration and differentiation [1]. The human FGF family has 18-22 members grouped into 6-7 subfamilies based on sequence homology and phylogeny [2]. The FGF family of growth factors have critical roles during both vertebrate and invertebrate embryonic development, in adult cells they modulate tissue maintenance, wound healing and angiogenesis [1,2].
The FGF ligands bind to 4 receptors (FGFR1-4), transmembrane receptors with intracellular tyrosine kinase activity [2, 3]. Once activated FGFRs recruit Src homology-2 (SH2) or phosphotyrosine binding (PTB) domain-containing signaling proteins leading to activation of intracellular signaling pathways [1]. The main signaling pathways activated by FGF binding are the RAS/MAP kinase pathway, PI3 kinase/Akt pathway, and PLCγ pathways [1-3].
Fibroblast growth factor 2 (FGF-2) also known as basic fibroblast growth factor (bFGF) has a broad range of physiological roles including regulation of cell growth, survival and proliferation. FGF-2 is one of the FGFs which binds to and signals through all four of the FGFRs [4].
FGF-2 is an essential growth factor for the maintenance of pluripotency in human embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs) [4] and mesenchymal stem cells (MSCs) [5]. FGF-2 preserves pluripotency through interaction with PI3 kinase/Akt, ERK1/2, JAK/STAT and PLCγ pathways by activation of activin A. In feeder culture systems FGF-2 stimulates production of essential growth factors and cytokines from the feeder layer [5].
FGF-2 and TGFβ1 are required for maintenance of iPSC in feeder free culture, Qkine produces high quality, bioactive and animal origin-free FGF-2 (Qk025) and TGF-β1 (Qk010) for stem cell culture. However, FGF-2 has poor thermostability in culture so, to prevent the need for daily media changes, Qkine FGF2-G3 has been engineered to be highly thermostable allowing weekend-free culture of iPSCs.
Additional resources
- Technote | FGF-2 145 aa (Qk025) bioactivity vs alternative supplier
- Technote | FGF-2 145 aa (Qk025) stability > 2 years lyophilized
- Technote | FGF-2 145 aa (Qk025) stability shipping conditions
- Technote | Tuna FGF-2 145 aa (Qk104) vs human FGF-2 145 aa (Qk025) bioactivity
- Differentiation of induced pluripotent stem cells (iPSCs) into neuroectoderm (PDF)
- Differentiation of induced pluripotent stem cells (iPSCs) into mesoderm (PDF)
- Qkine proteins are stable and bioactive after 14 days at room temperature and multiple freeze-thaws (PDF)
- Qkine lyophilized proteins are stable, bioactive and sterile after > 2 years in freezer storage (PDF)
- Poster: Neural and glial cell maintenance and differentiation
- Brochure: Growth factors for neural and glial cell differentiation
- Brochure: Growth factors for enhanced organoid culture protocols
Publications using Recombinant human FGF-2 (145 aa) protein (Qk025)
-
Hydrostatic pressure promotes chondrogenic differentiation and microvesicle release from human embryonic and bone marrow stem cells
Luo L, Foster NC, Man KL et al.
DOI: doi: 10.1002/biot.202100401 -
Refined and benchmarked homemade media for cost-effective, weekend-free human pluripotent stem cell culture
Truszkowski L, Bottini S, Bianchi S et al.
DOI: doi.org/10.12688/openreseurope.18245.2 -
Pervanadate-induced oxidation relieves autoinhibition of SRC protein tyrosine kinase
Mulholland KE, Bourguet M, Cheng N et al.
DOI: doi.org/10.1101/2025.04.27.650842
FAQ
Fibroblast growth factor 2 (FGF-2), also known as basic fibroblast growth factor (bFGF) is a growth factor and signaling protein.
FGF-2 is expressed in a developmental and tissue specific manner. It’s expression is tightly controlled in normal tissues and it can be detected in all major tissues.
FGF-2 is essential for normal embryonic development. It has roles in cell survival and proliferation, angiogenesis, tumorigenesis, wound healing and tissue repair.
FGF-2 binds to and signals though all four of the FGF receptors FGFR1-4.
FGFRs phosphorylate specific tyrosine residues and activate the RAS-MAPK, PI3K-AKT, PLCγ, and STAT intracellular signaling pathways.
FGF-2 is used to maintain the pluripotency of stem cells in culture.
We offer the 145 amino acid form rather than the 146 due to the E. coli processing of the N-terminal. When the 146 form (Pro143-Ser288) is expressed, the proline is either partially or completely removed. To improve expression in E. coli, we chose to start with the alanine (Ala144-Ser288) form. This modification does not affect receptor binding or bioactivity.
Other listings of the 146 amino acid form will often be a mixture of FGF-2 146 and 145, or, in some cases, only the 145 form.
-
FGF-2 (145 aa):
-
The most widely used form, supporting proliferation and survival of stem and progenitor cells.
-
Commonly preferred for organoid culture due to its robust activity and well-established performance.
-
-
FGF-2 (154 aa):
-
Contains an N-terminal extension that may affect intracellular localization or signaling.
-
Typically, not required for standard organoid or stem cell maintenance, unless specific signaling or localization effects are being investigated.
-
Recombinant FGF2-G3 (FGF2-STAB®) is a thermostable, engineered form of FGF-2 (bFGF). This modification significantly increases the protein’s functional half-life — from <10 hours for wild-type FGF-2 to >7 days for FGF2-G3 — improving stability and consistency in culture systems.
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.
For use in manufacturing of cellular or gene therapy products. Not intended for in vivo applications.

Receive a Qkine gift card when you leave us a review.
£100, $140 or €120 Qkine gift card for product reviews with an image and £50, $70 or €60 for reviews without an image.