Growth factors and cytokines for mouse and rat stem cell culture
Qkine is committed to manufacturing enhanced-quality bioactive proteins to improve scientific outcomes and reproducibility. We provide a complete portfolio of animal-origin free and carrier protein-free growth factors and cytokines for mouse and rat stem cell culture.
Maintenance of pluripotency in mouse stem cells
LIF is often regarded as the most important growth factor involved in mouse pluripotent stem cell maintenance. LIF, part of the IL-6 family, activates a number of pro-pluripotency signaling pathways including Jak-Stat3, PI3K-Akt, and YES-YAP. The JAK/STAT pathway regulates the core pluripotency regulatory circuit Oct4/Sox2/Nanog through activation of Klf4. PI3K-Akt blocks MAPK-Erk signaling, which is a driver of endoderm differentiation, and increases Tbx3 activity and Nanog expression. While the YES-YAP pathway induces Oct4 expression [1].
However, LIF also activates the MAPK-Erk signaling pathway, which promotes differentiation. To prevent this differentiation, two methods can be employed; inhibition of ERK using inhibitors such as PD0325901, this is the method employed in the popular 2i+LIF conditions, or activation of BMP signaling. BMP-4 inhibits MAPK and ERK signaling and regulates pluripotency gene expression through SMADs [1].
The third cytokine family shown to play a role in mouse pluripotency is the Wnt family. Activation of Wnt signaling stimulates self-renewal by inhibiting the repressor activity of endogenously expressed Tcf3. In the 2i+LIF conditions, inhibition of GSK3 activity (by CHIR99021) takes the place of Wnt signaling. Wnt3a has been shown to be capable of replacing GSK3 inhibition in mouse embryonic stem cell (mESC) culture and has been shown to sensitize cells to LIF [2].
Growth factors for mouse stem cell culture
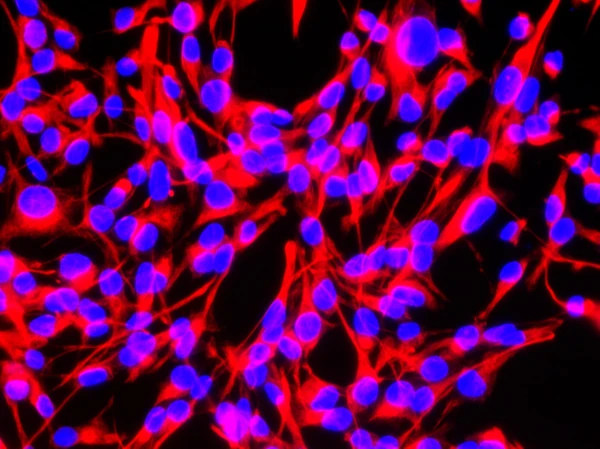
Expansion
Recombinant mouse/rat activin A protein (Qk012) Recombinant mouse/rat activin A PLUS™ protein (Qk014) Recombinant mouse EGF protein (Qk066) Recombinant mouse FGF-2 protein (Qk042) Recombinant mouse LIF protein (Qk018) Recombinant mouse noggin protein (Qk033) Recombinant mouse SCF protein (Qk077) Recombinant mouse TGF-β3 protein (Qk019)
Differentiation
Recombinant mouse/rat activin A protein (Qk012) Recombinant mouse/rat activin A PLUS™ protein (Qk014) Recombinant rat BDNF protein (Qk023) Recombinant mouse/rat BMP-2 protein (Qk009) Recombinant mouse/rat FGF-8a protein (Qk020) Recombinant mouse FGF-8b protein (Qk021) Recombinant rat FGF-10 protein (Qk016) Recombinant mouse LIF protein (Qk018) Recombinant mouse/rat NT-3 protein (Qk022)Human growth factors tested in mouse stem cells
| Homology human/mouse | Used in | |
|---|---|---|
| Recombinant human BMP-4 (Qk038) | 98% | Azami et al. [1], Drozd et al. [2] |
| Recombinant human follistatin-resistant activin A (FRACTA) (Qk035) | 98% | In house |
| Recombinant human GDF-15 (Qk017) | 67% | Fejzo et al. [3] |
| Recombinant human gremlin 1 (Qk015) | 99% | Sato et al. [4] |
| Recombinant human R-spondin 1 (Qk006) | 96% | Kirino et al. [5] |
| Recombinant human VEGF 165 (Qk048) | 88% | Azami et al. [1] |
Product focus
Recombinant mouse LIF is a cost-effective replacement for ESGRO
Mouse LIF (murine leukemia inhibitory factor) protein maintains the pluripotency and self-renewal of mouse embryonic and induced pluripotent stem cells.
Qkine recombinant mouse LIF protein is animal-free and carrier-protein free for highly reproducible results. Bioactivity was tested by colony formation assay and determination of Nanog expression.
Animal-free growth factors for mouse stem cell culture
We are confident that the quality and bioactivity of our recombinant proteins will ensure that you achieve your research goals. Our dedicated team of stem cell specialists is available to answer any queries you may have and to give expert support when required.

