Pioneering proteins
Qkine is committed to manufacturing enhanced-quality bioactive proteins to enhance scientific outcomes and improve reproducibility. We proactively leverage our expertise in manufacturing and protein engineering to develop unique, optimized proteins designed to address fundamental biological, translational, and scalability challenges.
Academic innovation combined with high purity manufacture
- Protein engineering from world-leading groups
- Licensed through technology transfer process
- Highest quality animal-free manufacture
- Stringent purity and bioactivity testing
- Adding value and innovation to stem cell science
Engineering novel functionality or specificity into growth factor proteins is a research time and resource intensive process. Successful new engineered proteins with unique properties of interest to stem cell science or regenerative medicine shouldn’t be retired to the back of the freezer.
To support translation of basic science, we are proud to launch our Pioneering Protein range; modified proteins developed by academic laboratories and manufactured to our high purity and bioactivity standards.
Are you interested in adding your own protein to the pioneering proteins range? Please contact us.
Pioneering proteins range
Follistatin-resistant activin A, FRACTA (Qk035)
Activin A, used widely in iPSC culture, is inhibited by follistatin; consequently activin A activity fluctuates in stem cell cultures. Each passage with fresh activin A containing media leads to high activin A activity, which declines as follistatin accumulates.
Follistatin-resistant Activin A (FRACTA) has the same bioactivity as wild-type activin A (Qk001) but is not affected by the natural feedback inhibition by follistatin.
This specialized activin A was developed in Dr Marko Hyvönen’s lab (University of Cambridge).

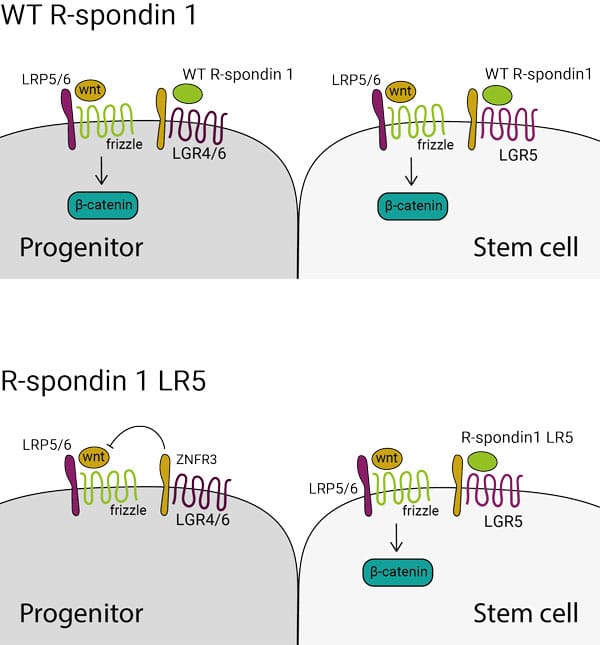
High affinity LGR5 receptor-binding R-spondin 1 LR5 (Qk031)
R-spondin 1 LR5 protein (Qk031) is a specialized form of R-spondin 1 that specifically binds to the LGR5 receptor, developed in the lab of Marc de la Roche, University of Cambridge.
R-spondin 1 (Qk006) is a ligand for the leucine-rich repeat-containing G-protein coupled receptors (LGR) 4-6. Binding of R-spondin 1 to its coreceptors, LGR4-6 and ZNRF3, releases the inhibition ZNRF3 imposes on wnt signalling.
In epithelial tissues LGR5 marks the stem cell population. This engineered form, R-spondin 1 LR5, activates wnt signalling only in the LGR5+
stem cell population.
R-spondin 1 LR5 support organoid survival and growth in intestinal organoid culture. As LGR5 specifically marks stem cells and is not found on transit amplifying cells, lower crypt multiplicity is seen in organoid cultures with R-spondin 1 LR5 (in comparison with WT R-spondin1), which supports the notion that this engineered form is acting specifically on stem cells.
Contact us
Our science team is here to help, please contact us if you have any questions.