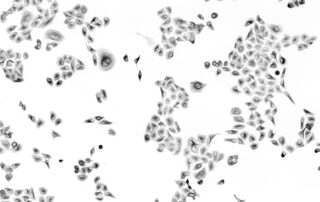
Qkine integrates the latest advancement in fill-finish automation into its recombinant protein manufacturing process
Our latest investment in our mission at Qkine to produce the highest quality growth factors for research. The most state-of-the-art automated system which fills, labels and caps vials with minimal manual intervention.