Raising the standard in bioactive protein
manufacturing and innovation
Qkine is committed to manufacturing bioactive proteins of the highest quality to enhance scientific outcomes and improve reproducibility.
Our robust animal-free manufacturing platform, along with rigorous quality control procedures, ensures exceptional bioactivity and consistent performance from lot to lot, guaranteeing outstanding performance in your applications. We proactively leverage our expertise in manufacturing and protein engineering to develop unique, optimized proteins designed to address fundamental biological, translational and scalability challenges.
We never compromise on the purity and bioactivity of our proteins. To ensure full reproducibility and enhance scientific outcomes, all our products adhere to the Nine-point Qkine Quality Commitment.

1. Certified animal origin-free with full traceability
- Compatible with serum-free and culture media for transition to clinical use and regulated applications, such as cellular agriculture
- Guaranteed absence of contamination from trace animal components or endogenous related proteins typically secreted during standard mammalian cell expression systems, ensuring highly defined culture conditions
- No risk of contamination from mammalian pathogens, free from BSE/TSE
2. Manufactured within our ISO 9001:2015-certified facility
with stringent in-process control measures
- Robust in-process controls and stage-gate analytics are employed to maximize lot reproducibility:
- Comprehensive protein expression parameter analysis conducted prior to refolding (if necessary) or purification
- Gel electrophoresis analysis used to validate proper refolding
- Chromatogram and analytical gel electrophoresis performed after each step of protein purification chromatography, compared against documented standards
- Thorough oversight of the entire process and manufacturing procedure is maintained through the Qkine bespoke ISO 9001:2015 quality management system
- Experimentally determined shelf-life and shipping stability analysis ensure the protein maintains its reproducible bioactivity during global transit to our customers
- Detailed, lot-specific certificates of origin (CoO) and certificates of analysis (CoA) are provided
- The custom design of our UK manufacturing facility includes provisions for beta-lactam-free production. No baculovirus is handled on-site

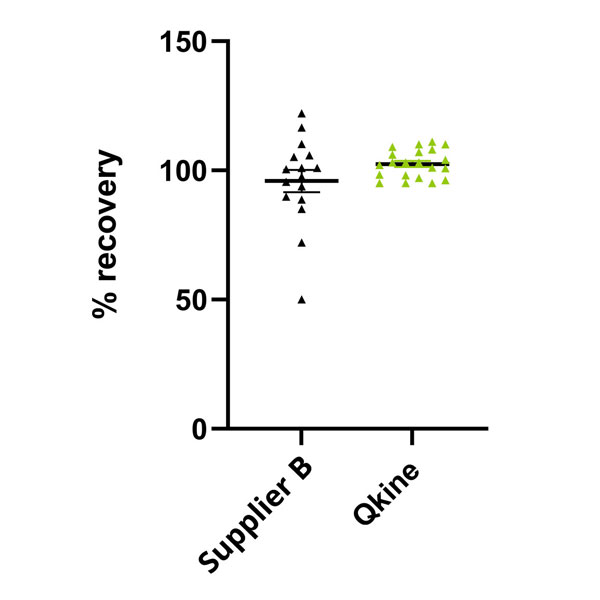
3. Rigorously tested for accurate protein recovery from vials
- Protein recovery from sample vials in each lot is precisely quantified using UV spectrophotometry, ensuring compliance with our industry-leading vial recovery standard of 95%-115%
- As part of our research and development process, we experimentally define the correct reconstitution buffer tailored to each specific protein. We provide this reconstitution solution at no additional cost to our customers, simplifying laboratory workflows and media preparation
- Accurate protein recovery assurance empowers our customers to confidently utilize precise final concentrations of bioactive proteins in their applications, promoting both inter and intra-laboratory reproducibility and minimizing wastage of time and resources
4. Subject to quantitative bioactivity analysis with integrated lot-to-lot reproducibility criteria
- Validated protein-specific quantitative luciferase reporter assay or proliferation assay to determine bioactivity accurately
- Bioactivity assay performed on every lot, and rigorous direct lot-to-lot comparison on the same assay plate is built into the quality control and manufacturing lot releases standard operating procedures
- If the protein is not statistically comparable with the representative lot, it is discarded
5. Proven to have industry-leading protein purity levels
- We routinely perform SDS gel electrophoresis on 3 µg and 7 µg aliquots of all our recombinant proteins under both reduced and non-reduced conditions. This standard practice for every manufacturing lot ensures the absence of contaminants, aggregates, or degradation products
- While we believe our proteins exhibit purity levels exceeding 99.9%, as indicated by mass spectrometry analysis, the inherent limitations of gel staining and densitometry introduce a margin of error of +2%. Consequently, we report purity at the limit of the assay, which is greater than 98%
6. Subject to comprehensive identity analysis for structurally complex bioactive proteins
- Each batch of protein undergoes meticulous analysis via sensitive mass spectrometry (LC-MS) to confirm the molecular mass of the intact protein, definitively establishing its identity
- For proteins like those in the TGF-beta family, where the active form is the dimeric protein, we confirm the protein is the pure dimeric form
- This process reveals any potential heterogeneity or trace contaminants that may not be discernible through SDS-PAGE analysis
7. Assayed to ensure industry-leading low endotoxin levels
- While industry-standard endotoxin levels in recombinant growth factors are often <0.5 EU/ µg, our internal standards are more stringent, requiring levels of <0.1 EU per µg of protein
- The majority of Qkine growth factors exhibit endotoxin levels below the limit of detection (<0.0005 EU/μg protein in standardised FDA-compliant assays). Interestingly, we have also benchmarked mammalian-expressed proteins, which should theoretically contain no endotoxin, and measured endotoxin levels exceeding the threshold of 0.25 EU/ μg
8. Rigorously tested for sterility and mycoplasma contamination for confident use
in cell-culture applications
- Products are thoroughly screened for mycoplasma presence using two distinct assays:
a) Detection of enzymes present in nearly all of the approximately 200 mycoplasma species but absent in eukaryotic cells (via luminescence)
b) PCR-based testing targeting the conserved 16S ribosomal RNA coding region within the Mycoplasma genome - Sterility testing is conducted under both aerobic and anaerobic conditions to identify potential contaminants such as fungi, yeast, and bacteria, ensuring total confidence in cell-culture applications
9. From concept to manufacture, our proteins are designed to be the best
- Where it is required to address scientific challenges, we use protein engineering to enhance specific protein properties, such as stability in culture media or manufacturing efficiency, whilst retaining full biological activity and specificity. In some cases, we can engineer the proteins to enhance biological activity, as seen with follistatin-resistant Activin A where we have removed natural feedback inhibition to allow fine-tuning of culture conditions
- The final form of every protein is free from protein tags such as His-tags to remove scientific uncertainty and ensure compatibility with translational studies
- We design quality into every aspect of our protein manufacturing processes and the proteins themselves so you can have total confidence in their efficacy in your applications. Higher-compliance quality assurance, bespoke QA services and regulatory documentation are available on request
Getting the simple things right is important.
Qkine is a scientist-led company committed to developing and manufacturing the highest quality bioactive proteins to improve scientific outcomes, enhance reproducibility, and reduce wasted time and budget. We achieve this through our dedication to scientific excellence, integrity, and continual improvement.
Our science team is here to help, please contact us if you have any questions at customerservice@qkine.com.