

Price range: £250.00 through £3,190.00
Activin A is a TGF-β family growth factor regulating embryonic development, cell proliferation, differentiation, and immune responses. Activin A is frequently used to maintain pluripotency in induced pluripotent and embryonic stem cell cultures. It is also used in many stem cell differentiation protocols, including endoderm lineage differentiation and further maturation into hepatocyte and pancreatic cells.
Recombinant human activin A protein is a high-purity mature bioactive dimer of 26 kDa. It is animal origin-free (AOF), carrier protein-free, and tag-free to ensure its purity with exceptional lot-to-lot consistency. This protein has been rigorously benchmarked against other commercial sources and extensively validated for highly reproducible stem cell culture. Qkine founder Marko Hyvönen developed the protocol for making high-purity activin A, and this protein is used at the Cambridge Stem Cell Institute.
High purity mature bioactive dimer rigorously benchmarked against other commercial sources.
Qkine activin A is also available as cell therapy grade with extended quality testing and documentation – Qk001-CTG
In stock
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
Price range: £250.00 through £3,190.00
Buy online with secure credit card or purchase order. For any questions, please email orders@qkine.com
>98%, by SDS-PAGE quantitative densitometry
Expressed in E. coli
Animal origin-free (AOF) and carrier protein-free
Manufactured in our Cambridge, UK laboratories
Lyophilized from acetonitrile, TFA
Induced pluripotent and embryonic stem cell differentiation and maintenance
iPSC-derived mesoderm differentiation
Differentiation of iPSC into endoderm
Chemically defined media for organoid culture
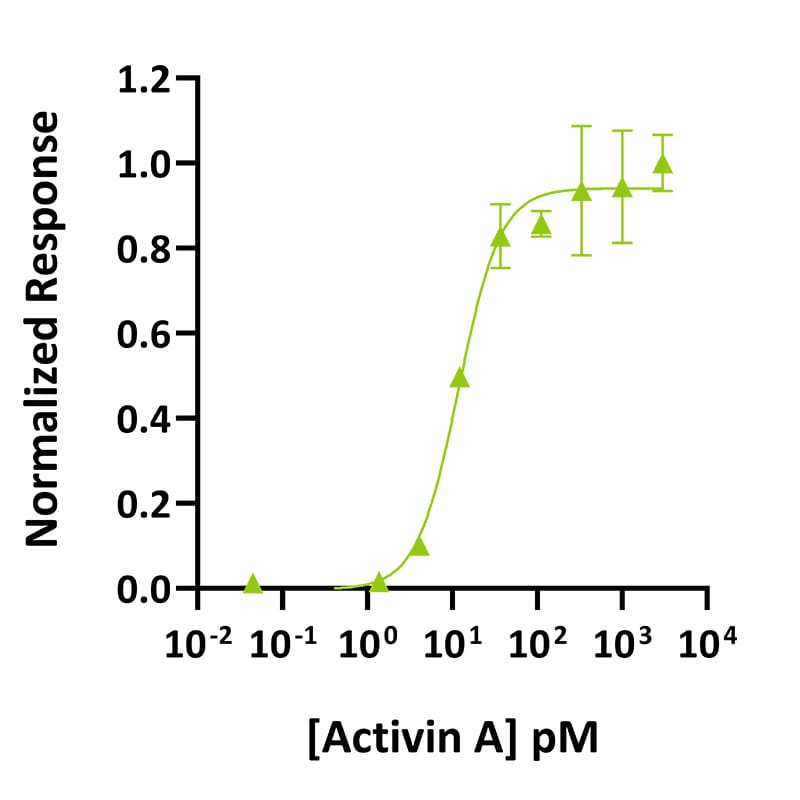
Activin A activity was determined using the activin-responsive firefly luciferase reporter assay in transiently transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of activin A. Firefly luciferase activity was measured and normalized. EC50 = 11.9 pM (309.4 pg/ml). Data from Qk001 lot #104276.
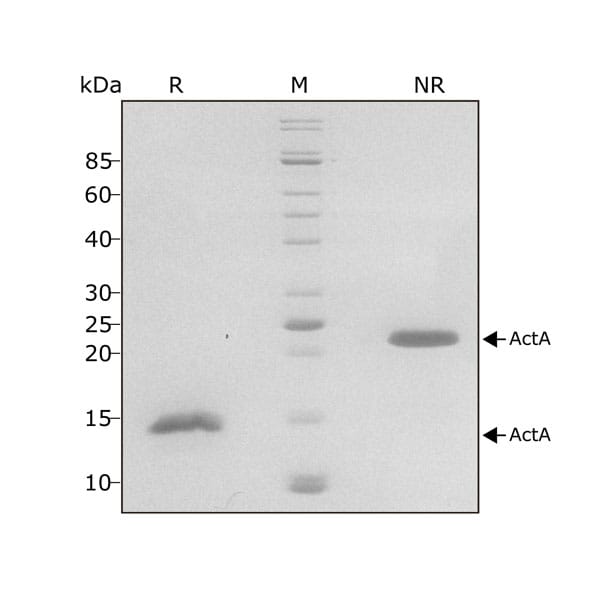
Activin A migrates as a single band at 24 kDa in non-reducing (NR) and 13 kDa as a single monomeric species upon reduction (R). No contaminating protein bands are visible. Purified recombinant protein (7 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptothanol, R) and non-reduced conditions (NR) and stained with Coomassie Brilliant Blue R250. Data from Qk001 lot #011.
Mass spectrometry: single species with expected mass
Recovery from stock vial: >95%
Endotoxin: <0.05 EU/μg protein
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.

Qkine activin A was as bioactive as activin A protein from an alternative supplier. Activin A activity was determined using the activin-responsive firefly luciferase reporter assay in transiently transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of Qkine activin A (Qk001, green) or alternative activin A (Supplier B, black). Firefly luciferase activity was measured and normalized. Data from Qk001 lot #204536 EC50 = 16 pM, Supplier B EC50 = 12 pM.
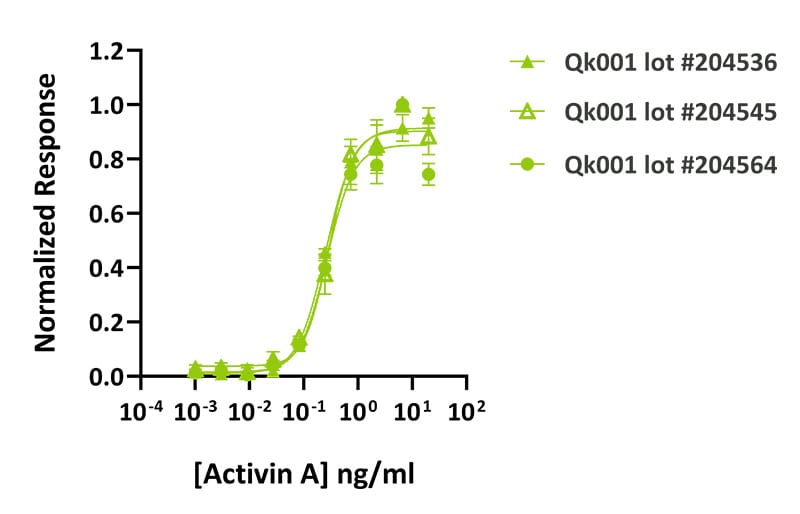
Qkine activin A had consistent bioactivity over 3 independently manufactured lots. Activin A activity was determined using the activin-responsive firefly luciferase reporter assay in transiently transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of 3 independent lots of Qkine activin A. Firefly luciferase activity was measured and normalized. Lot #204536 EC50 = 0.26 ng/ml, lot #204545 EC50 = 0.29 ng/ml, lot #204564 EC50 = 0.27 ng/ml.
Activin A is a member of the belongs to the transforming growth factor beta (TGF-β) superfamily TGF-β family of growth factors [1–4]. It was first identified for its ability to stimulate the release of follicle-stimulating hormone (FSH) from the pituitary gland. It was later recognized as a multifunctional protein with diverse cellular effects [5, 6]. It plays crucial physiological roles in regulating embryonic development, cell proliferation, and differentiation [6, 7]. It promotes the patterning and differentiation of various organs, including the development of the mesoderm, neural, and reproductive systems. It is also involved in maintaining homeostasis, regulating immune responses, and wound healing [8]. Impaired activin A signaling has been associated with various pathological conditions, including cancer, inflammation, and fibrosis [9, 10]. As activin A can contribute to disease progression and severity, it is a growing area of research for promising therapeutic targets.
Activins are disulfide-linked homo- and heterodimers of four inhibin β chains [6]. The best-characterized are activin A and activin B, homodimers of inhibin βA and inhibin βB, respectively. Like all other members of the TGF-β family, activins are synthesized as significant precursors consisting of an N-terminal signal peptide, a pro-domain of 250–350 residues, and a highly conserved mature domain. The pro-domain, which is cleaved off in the mature protein, has essential roles in the biosynthesis, stabilization, transportation and signaling of the growth factors in the body [11]. Activin A binds to activin type I (ALK4 or ALK7) and type II (ActRIIA or ActRIIB) receptors [6, 12]. The type II receptor phosphorylates the type I receptor upon ligand binding, initiating downstream signaling cascades, mainly through the SMAD family of proteins. Activated SMAD complexes translocate into the nucleus, regulating the transcription of target genes involved in various cellular processes. In vivo, the high-affinity inhibitor follistatin and inhibins tightly regulate activin A activity in a feedback loop [13, 14]. Follistatin is secreted into the media during stem cell culture. However, the impact on the efficiency of stem cell differentiation and cellular homogeneity has not been studied closely (see this discussion for more information).
Activin A is frequently used for the maintenance of pluripotency of human induced pluripotent stem cells and human embryonic stem cell lines along with fibroblast growth factor 2 (FGF-2) [2, 4]. Activin A is also used in various stem cell differentiation protocols. It directs the differentiation into definitive endoderm, precursor to different cell types such as pancreatic and liver cells [15–18]. Activin A also promotes neural precursor cells and drives astrocytic differentiation with Ciliary neurotrophic factor (CNTF) and Glial cell line-derived neurotrophic factor (GDNF) [19]. Moreover, activin A is also involved in mesodermal differentiation to derive muscle, bone, and blood cells. Finally, activin A is often used for the development and maintenance of organoids [18, 20, 21]. Qkine recombinant activin A protein has been extensively validated and benchmarked with other suppliers’ proteins in stem cell culture and assays. You can view the results of this analysis and a commentary by Qkine’s founder, Marko Hyvönen.
Andreasson L, Evenbratt H, Mobini R and Simonsson S et al.
DOI: doi: 10.1016/j.jbiotec.2020.10.030Blackford SJI et al.
DOI: doi.org/10.1002/sctm.18-0084Stuart HT et al.
DOI: doi: 10.1016/j.stem.2019.07.009Wamaitha SE, Grybel KJ, Alanis-Lobato G et al.
DOI: doi: 10.1038/s41467-020-14629-xBoikova A, Quaife-Ryan GA, Batho CAP et al.
DOI: https://doi.org/10.1101/2023.08.02.551469Barsby T, Ibrahim H, Lithovius V et al.
DOI: doi: 10.1016/j.xpro.2022.101711.Weatherbee BAT, Weberling A, Gantner CW et al.
DOI: doi: 10.1038/s41556-024-01367-1Jobbins AM, Haberman N, Artigas N et al.
DOI: doi: 10.1093/nar/gkac165Carbognin E, Carlini V, Panariello F et al.
DOI: doi: 10.1038/s41556-023-01131-xGang N
DOI: ThesisFarhan F, Trivedi M, Di Wu P et al.
DOI: doi: 10.1186/s13287-023-03542-xRossignoli G, Oberhuemer M, Brun IS et al.
DOI: https://doi.org/10.1101/2025.01.17.633522Darrigrand J-F, Isaacson A and Spagnoli FM
DOI: doi: 10.12688/f1000researchJalil S, Keskinen T, Juutila J et al.
DOI: doi: 10.1016/j.ajhg.2024.03.004Luo L, Foster NC, Man KL et al.
DOI: doi: 10.1002/biot.202100401Miguel-Escalada I, Maestro MÁ, Balboa D et al.
DOI: doi: 10.1016/j.devcel.2022.07.014Weatherbee BAT, Gantner CW, Iwamoto-Stohl LK et al.
DOI: doi: 10.1038/s41586-023-06368-yRosa VS, Sato N and Shahbazi MN et al.
DOI: doi: 10.1016/j.xpro.2024.103347Militi S, Nibhani R, Jalali M and Pauklin S.
DOI: doi: 10.1016/j.celrep.2023.113146Truszkowski L, Bottini S, Bianchi S et al.
DOI: doi.org/10.12688/openreseurope.18245.2Farbergshagen, AC (Thesis)
DOI: ThesisGuo M, Wu J, Chen C et al.
DOI: doi: 10.1038/s41467-024-44969-xBalmas E, Ratto ML, Snijders KE et al.
DOI: http://dx.doi.org/10.2139/ssrn.4854180Azami T, Theeuwes B, Ton M-LN et al.
DOI: doi: 10.1016/j.celrep.2025.115506Meek S, Watson T, Eory L et al.
DOI: doi: 10.1186/s12915-021-01217-8Bao M, Cornwall-Scoones J, Sanchez-Vasquez E et al.
DOI: doi: 10.1038/s41556-023-01157-1Beucher A, Miguel-Escalada I, Balboa D et al.
DOI: doi: 10.1038/s41556-022-00996-8Zorzan I, Pellegrini M, Arboit M et al.
DOI: doi: 10.1038/s41467-020-16205-9Garitta E
DOI: https://qmro.qmul.ac.uk/xmlui/handle/123456789/105833Bernardo, Edgar et al.
DOI: 10.1016/j.cmet.2025.07.007Chen C, Wu J, Wang X et al.
DOI: DOI: 10.1016/j.stem.2025.08.011Becca S, Bianchi S, Hahn EM et al.
DOI: doi.org/10.1101/2025.10.07.680441Azami T, Patton EE and Nichols J.
DOI: doi.org/10.1101/2025.10.15.682610Williams TL, Macrae RGC, Kuc RE, Brown AJH, Maguire JJ, Davenport AP.
DOI: doi: 10.3389/fendo.2023.1139121Keskinen T, Jalil S, Gümüşoğlu I et al.
DOI: doi: 10.1002/jimd.70122Skowronska-Krawczyk et al.
DOI: doi.org/10.21203/rs.3.rs-8607320/v1Zamarian V, Monaco L, Marras M et al.
DOI: doi.org/10.1007/s12015-026-11091-yBottini S (Thesis)
DOI: ThesisRossignoli G, Oberhuemer M, Brun IS et al.
DOI: https://doi.org/10.1038/s44318-026-00714-2Hernanz M, Matas D, Mirasierra M et al.
DOI: DOI: 10.3791/69562Activin A is a multifunctional protein. It is involved in regulating embryonic development, cell proliferation, and differentiation. It promotes the patterning and differentiation of various organs, including the development of the mesoderm, neural, and reproductive systems. It is also involved in maintaining homeostasis, regulating immune responses, and wound healing. Finally, it stimulates the release of follicle-stimulating hormone from the pituitary gland.
Activin A is a critical factor in stem cell culture, commonly used to maintain the pluripotency of induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs). It is integral to various stem cell differentiation protocols, guiding the differentiation of the definitive endoderm, neural and mesodermal lineages. Activin A is also commonly utilized in the development and maintenance of organoids.
Activin A and inhibin A are closely related proteins in the TGF-β superfamily. They share structural similarities but have distinct functions and roles in regulating various physiological processes. Activin A has broader functions beyond reproduction, which include handling embryonic development, stem cell maintenance, and the immune system. Inhibin A, conversely, is more specifically associated with the feedback control of follicle-stimulating hormones in the context of reproductive physiology.
Activin A binds to activin type I (ALK4 or ALK7) and type II (ActRIIA or ActRIIB) receptors to activate downstream SMAD signalling.
Yes, activin is involved in a feedback loop that regulates the secretion of follicle-stimulating hormone (FSH) and luteinising hormone (LH).
The activin gene family includes several genes that encode different activin subunits, forming various activin isoforms such as Inhibin Beta Subunits (INHBA / INHBB / INHBC / INHBD).
Follistatin and inhibins tightly regulate activin A activity in a feedback loop.
TGF beta family proteins and other growth factors can be very poorly soluble in physiological solutions. Please follow the handling guidance for lyophilized cytokines below to minimize loss of protein due to precipitation or adsorption to plastic. We advise storing the recombinant protein at very low pH before dilution in cell culture media or final working solutions. Low pH will also assist in maintaining the correct disulfide structure of the protein by minimizing disulfide bond exchange reactions.
Every effort is made to ensure samples are sterile; however, we recommend sterile filtering after dilution in media or the final working solution.
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.
For use in manufacturing of cellular or gene therapy products. Not intended for in vivo applications.

£100, $140 or €120 Qkine gift card for product reviews with an image and £50, $70 or €60 for reviews without an image.

