Currency
Recombinant human/bovine/porcine TGF-β1 PLUS ™ protein (Qk010)
Transforming growth factor-beta 1 (TGF-β1) is a pleiotropic cytokine that regulates various cellular processes, including cell proliferation, growth, differentiation, motility, and apoptosis. It is an essential growth factor in many embryonic and induced pluripotent stem cell maintenance media, including the commonly used E8, StemPro, and mTeSR media. TGF-β1 also promotes the differentiation of various cell types such as fibroblasts, epithelial cells, and immune cells.
Human/bovine/porcine recombinant TGF-β1 PLUS protein is the first entirely animal origin-free recombinant human TGF-β1 protein for highly reproducible results and compatible with chemically-defined stem cell media.
TGF-β1 PLUS is a high purity 24 kDa dimer comprising optimized mature domain of TGFβ1 protein. Our TGF-β1 PLUS protein has been extensively tested for maintenance of iPSC pluripotency by the specialist stem cell biotechnology company, Stemnovate, Cambridge, UK.
*Benchmark our TGF-β1 PLUS against your existing supplier*
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
1000µg will be despatched as 2 x 500µg
Fast and free shipping.
Buy online with secure credit card or purchase order.
For any questions, please email orders@qkine.com
Summary
High purity optimised human/bovine/porcine TGF-β1 protein (Uniprot: P01137)
>98%, by SDS-PAGE quantitative densitometry
24 kDa (dimer)
Expressed in E. coli
Animal-free (AOF) and carrier protein-free.
Manufactured in our Cambridge, UK laboratories
Lyophilized from acetonitrile, TFA
Resuspend in 10mM HCl at >100 µg/ml (provided with protein and free of charge), prepare single use aliquots, add carrier protein if desired and store frozen at -20°C or -80°C
Featured applications
iPSC and ESC maintenance and expansion
Chemically defined media
Human, bovine, porcine
Species similarity:
Mouse – 99%
Rat – 99%
Related discovery kit
Bioactivity
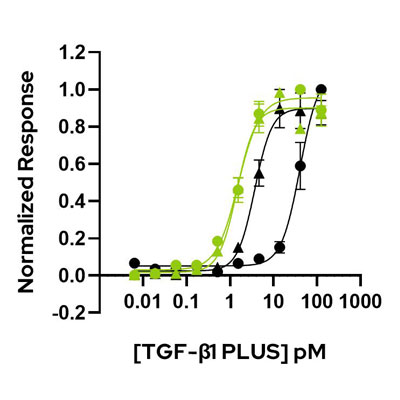
TGF-β1 PLUS (Qk010) is highly bioactive compared to mammalian-expressed TGF-β1 preparations from other suppliers.
Comparative activity was determined using a quantitative luciferase reporter assay in transiently transfected HEK293T cells. Cells were treated (in triplicate) with a serial dilution of TGF-β1. Firefly luciferase activity is measured and normalized to control Renilla luciferase activity. Qk010 TGF-β1 PLUS lot #011 and #012 (green) both have an EC50 of 1.45 and 1.54 pM (~36 pg/mL). Suppliers 1 and 2 (black) have EC50 of 3.7 pM and 40.7 pM respectively. Data from Qk010 lot #011 and #012.
Purity
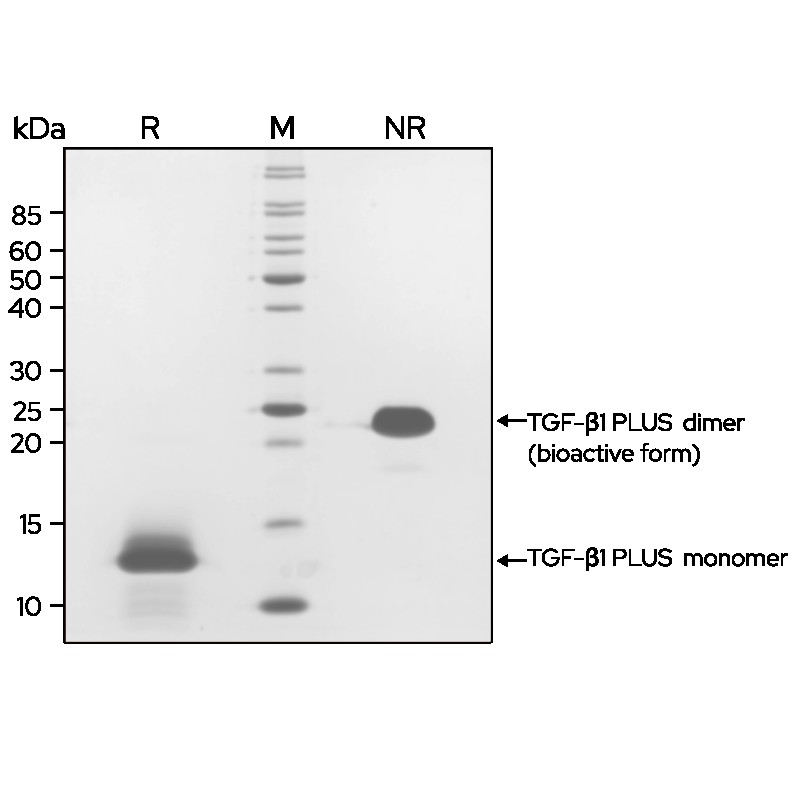
TGF-β1 PLUS (Qk010) dimer migrates as a single band at 24 kDa in non-reducing (NR) and 13 kDa as a single monomeric species upon reduction (R). High purity yield of dimeric protein (bioactive form).
Purified recombinant protein (7 μg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptoethanol, R) and non-reduced conditions (NR) and stained with Coomassie Brilliant Blue R250. Data from Qk010 lot #012.
Further quality assays
Mass spectrometry: single species with expected mass
Analytical reversed-phase: single sharp peak
Endotoxin: <0.005 EU/μg protein (below level of detection)
Recovery from stock vial: >95%
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.
Protein background
Transforming growth factor-beta 1 (TGF-β1) is a pleiotropic cytokine part of the TGF-β superfamily. TGF-β1 regulates various cellular processes, including cell proliferation, growth, differentiation, motility, and apoptosis [1]. It plays a crucial role in the immune response, tissue repair, and the epithelial-mesenchymal transition. Transforming growth factor-beta 1 is produced by various cell types, including immune cells, fibroblasts, and epithelial cells. It is synthesized and secreted as an inactive or latent complex, associated with latency-associated proteins (LAP), and targeted to the extracellular matrix [2]. It is released from latency by TGF-β activators including plasmin, matrix metalloproteases, integrins… Once the LAP cleaved, the mature transforming growth factor-beta 1 is a homodimeric protein composed of two identical subunits linked by a disulfide bond. Its amino acid sequence is composed of 390 amino acids. TGF-β1 signals through complexes of cell surface receptors including TGF-βRII/TGF-βRI and ALK-5/ALK-1. This triggers downstream signaling cascades, such as the Smad-dependent and Smad-independent pathways.
In cell culture, recombinant TGF-β1 protein is an essential growth factor in many embryonic and induced pluripotent stem cell maintenance media, including the commonly used chemically-defined E8, StemPro, and mTeSR medias [3–5]. Transforming growth factor-beta 1 supports the survival and maintenance of pluripotency of stem cells [1]. TGF-β1 is used to promote the differentiation of various cell types such as fibroblasts, epithelial cells, and immune cells. It is used in combination with other growth factors such as BMP-2 to regulate bone marrow stromal cell differentiation or with IL-2 and IL-6 to regulate T reg and Th17 cells differentiation [6–8].
To date, recombinant human TGF-β1 has only been produced from mammalian cell protein expression systems (HEK or CHO), where endogenous protein contaminants, cost and animal-free status is a challenge. As part of the ongoing mission to redefine industry standards for growth factor and cytokine biochemical quality, Qkine introduced the first optimized, animal-free, and highly bioactive recombinant human TGF-β1.
Benefits:
- Truly animal-free alternative for chemically-defined stem cell culture media
- High potency in iPSC culture with high pluripotency (Nanog) marker expression
- High purity, extensive biochemical data, and exceptional batch-to-batch consistency
- What does TGF-beta1 do?
TGF-β1 is a multifunctional cytokine that plays a crucial role in various cellular processes. It regulates cell growth, differentiation, proliferation, migration, and apoptosis. It is also involved in the immune response, epithelial-mesenchymal transition, and the extracellular matric synthesis and modelling.
- What kind of protein is TGF-beta?
It is a member of the transforming growth factor-beta superfamily of cytokines that are synthesized as latent complexes. TGF-β1 requires further processing to become the biologically active, mature TGF-β1.
- What disease is associated with TGF-beta1?
Dysregulation of TGF-β1 signaling is associated with various diseases such as cancer, fibrosis, autoimmune, cardiovascular, neurological, and chronic kidney diseases.
- What does a high human TGF-beta1 mean?
Elevated levels of TGF-β1 in the human body can have different implications such as higher risks of fibrosis, cancer progression, chronic inflammation, and cardiovascular diseases. It is important to consider the specific clinical context, the presence of other biomarkers, and the patient’s prognosis.
- What activates TGF-beta?
TGF-β1 is synthesized and secreted in an inactive or latent form. It is activated through cleavage and binding to matrix metalloproteinases, integrins, extracellular matrix such as thrombospondin-1, but also pH changes and reactive oxygen species.
Customer & collaborator data
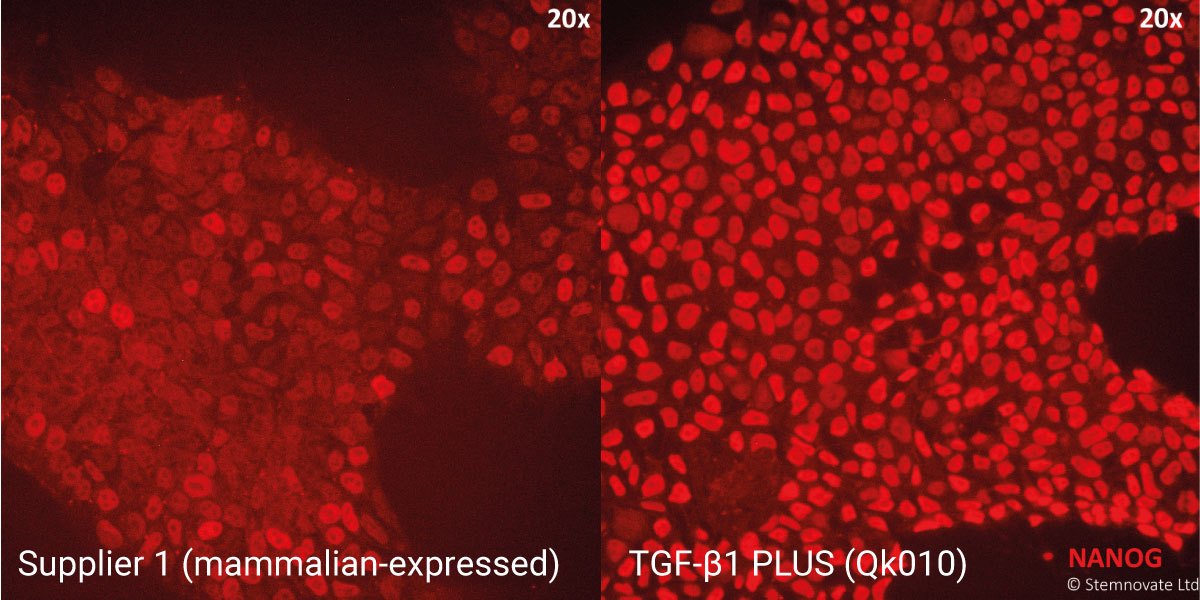
TGF-β1 PLUS maintains iPSC pluripotency with high Nanog expression
TGF-β1 PLUS is highly effective at maintaining iPSC pluripotency in chemically-defined, serum and feeder-free culture
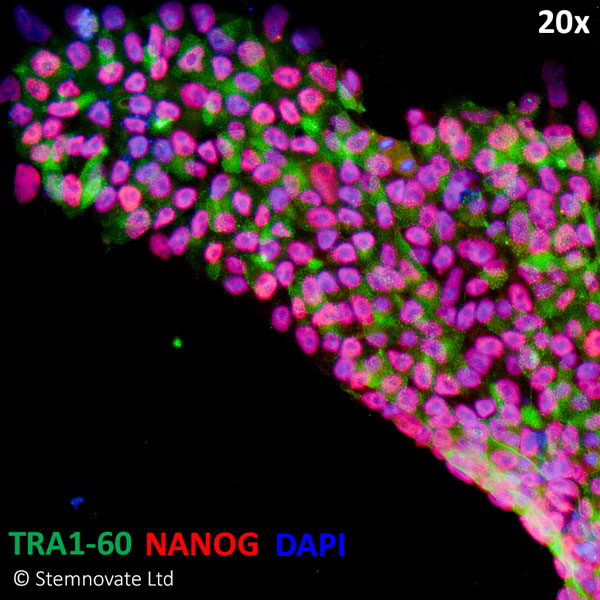
Immuno-staining for pluripotency markers Tra 1-60 and Nanog show high levels of expression in iPSC lines maintained in TGF-β1 PLUS (Qk010)-containing defined media. In this study, Qkine TGF-β1 PLUS performed better than the TGF-β1 used routinely by Stemnovate.
TGF-β1 PLUS (Qk010) maintains pluripotency and good colony morphology at 1 ng/ml in a chemically-defined serum and feeder-free iPSC culture. TGF-β1 PLUS used at 1 ng/ml; Qk025 FGF2 used at 100 ng/ml.
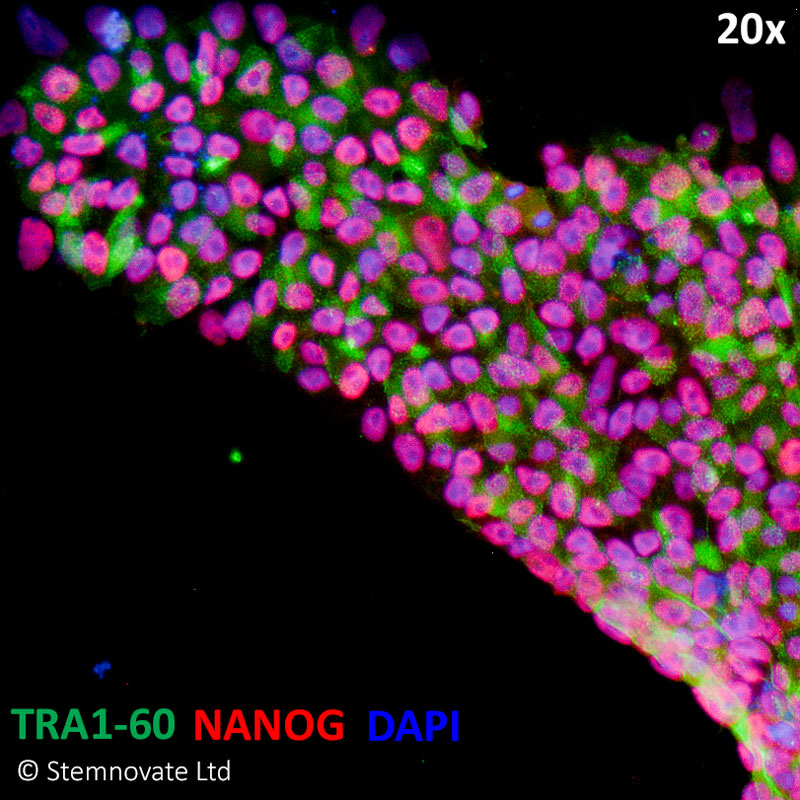
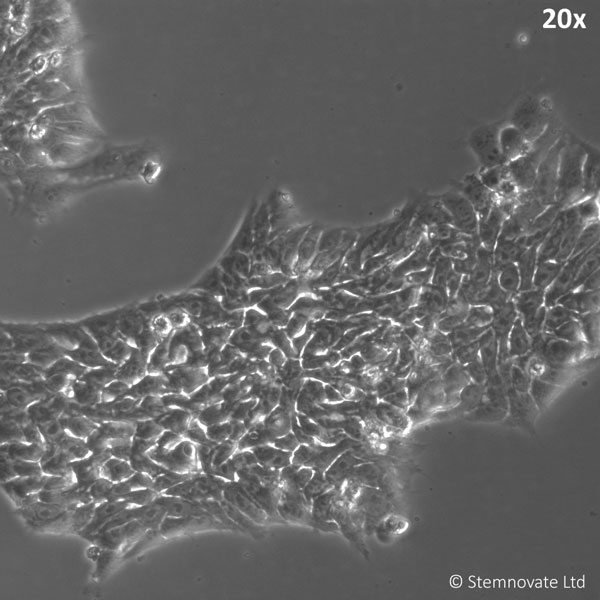
TGF-β1 PLUS in combination with Qkine FGF2 shows enhanced bioactivity when benchmarked against other suppliers
Stemnovate IPS media (chemically-defined, serum and feeder-free culture). Higher Nanog pluripotency marker immuno-staining in human iPSC cultured in Qkine FGF2 (Qk025) and TGF-β1 PLUS (Qk010).TGF-β1 PLUS used at 1 ng/ml.
All experiments have been conducted by the specialist stem cell biotechnology company, Stemnovate Limited, in Cambridge, UK.
Publications
Cytotherapy. 2024
Modeling the selective growth advantage of genetically variant human pluripotent stem cells to identify opportunities for manufacturing process control.
Authors: Beltran-Rendon, C., Price, C. J. et al.
Labs: Robert Thomas, Loughborough University
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.
Share your thoughts!
Let us know what you think...
What others are saying



Question
Anonymous –
I have read your FAQ on protein handling. Still, resuspending proteins in low pH 10mM HCl is unusual for most proteins but apparently normal for TGF-Beta. Why is this the case for TGF-beta and would resuspending in pure water or PBS be acceptable for short-term storage? Thanks.
Upvote if this was helpful (0) Downvote if this was not helpful (0) Flag for removal
Mairi Shepherd –
TGF-beta family proteins are very hydrophobic and consequently are poorly soluble and will adhere to plastic, we highly recommend following the handling guidance for both long- and short-term storage. We do not recommend resuspending in water or PBS as TGF-beta is insoluble in physiological buffer and the protein will precipitate. We advise storing the protein at a low pH to ensure solubility before dilution to cell culture media. A low pH will also aid the maintenance of the correct disulphide structure of the protein by minimising disulphide bond exchange reactions.
To save you the trouble of making a 10mM HCl solution, we ship vials of reconstitution solution A (10mM HCl) with proteins for which this is our recommended reconstitution buffer. Alternatively, you can make your own 1:1000 dilution of concentrated HCl. If you have any questions or would like to arrange a call with our protein team to discuss your concerns, please email support@qkine.com.
Upvote if this was helpful (1) Downvote if this was not helpful (0) Flag for removal