Recombinant human FGF-2 (bFGF) 154 aa protein (Qk027)
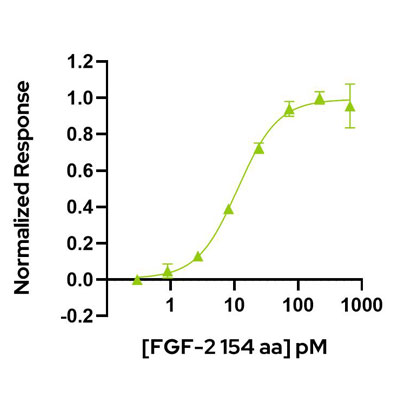
Recombinant human FGF2 protein (154 aa) is a highly bioactive, long–form of human fibroblast growth factor 2 protein. FGF-2 protein is used to support the maintenance of human embryonic stem cells and proliferation and differentiation of induced pluripotent and mesenchymal stem cells. This 154 aa form of FGF-2 comprises the core structured region and N-terminal extension.
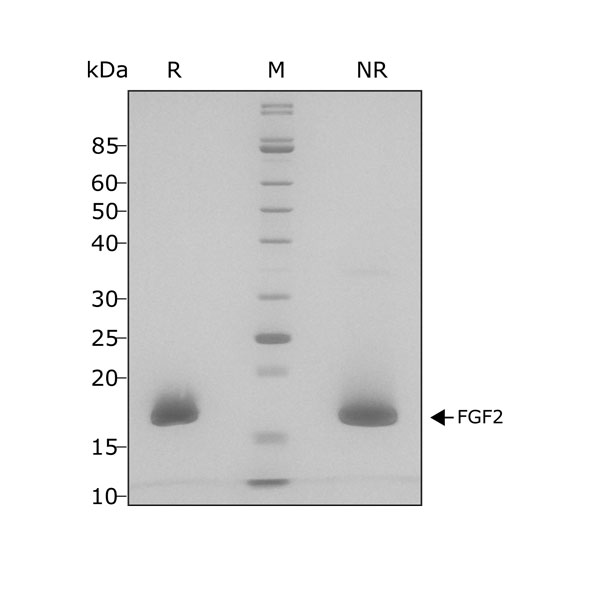
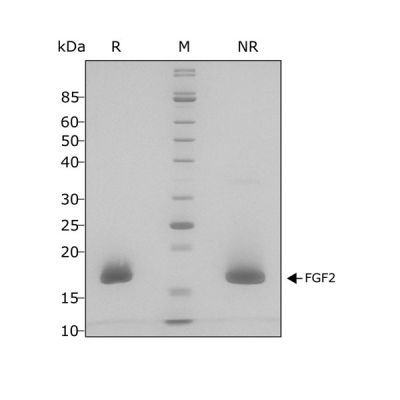
High purity and bioactivity 17 kDa FGF-2 / bFGF protein, animal-free (AOF) and carrier-protein free (CF).
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.


What others are saying
There are no contributions yet.