

Price range: £190.00 through £2,090.00
R-spondin 1 protein (RSPO1) is the prototypic member of the R-spondin family and is used to potentiate Wnt signaling in many organoid culture systems including intestinal and tumor (cancer) organoid culture. R-spondin 1 is also required for hematopoietic stem cell specification and cancer cell migration and survival.
Recombinant human R-spondin 1 protein (RSPO1) protein is the bioactive domain of human R-spondin 1 comprising the two cysteine-rich furin-like domains of R-spondin 1. Those are necessary and sufficient for Wnt signaling potentiation and are the essential domains for activity in stem cell and organoid culture.
This protein has a molecular weight of 13 kDa and is animal origin-free (AOF), carrier protein-free, and tag-free to ensure its purity with exceptional lot-to-lot consistency. Qk006 is suitable to replace R-spondin conditioned media for improved reproducibility in chemically defined organoid culture media.
This protein is also available as GMP compliant Cell Therapy Grade, to enquire email support@qkine.com.
In stock
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
Price range: £190.00 through £2,090.00
Buy online with secure credit card or purchase order. For any questions, please email orders@qkine.com
>98%, by SDS-PAGE quantitative densitometry
Expressed in E. coli
Animal origin-free (AOF) and carrier protein-free
Manufactured in our Cambridge, UK laboratories
Lyophilized from acetonitrile, TFA
Pancreatic tumor organoid culture
Maintenance of urine-derived stem cells
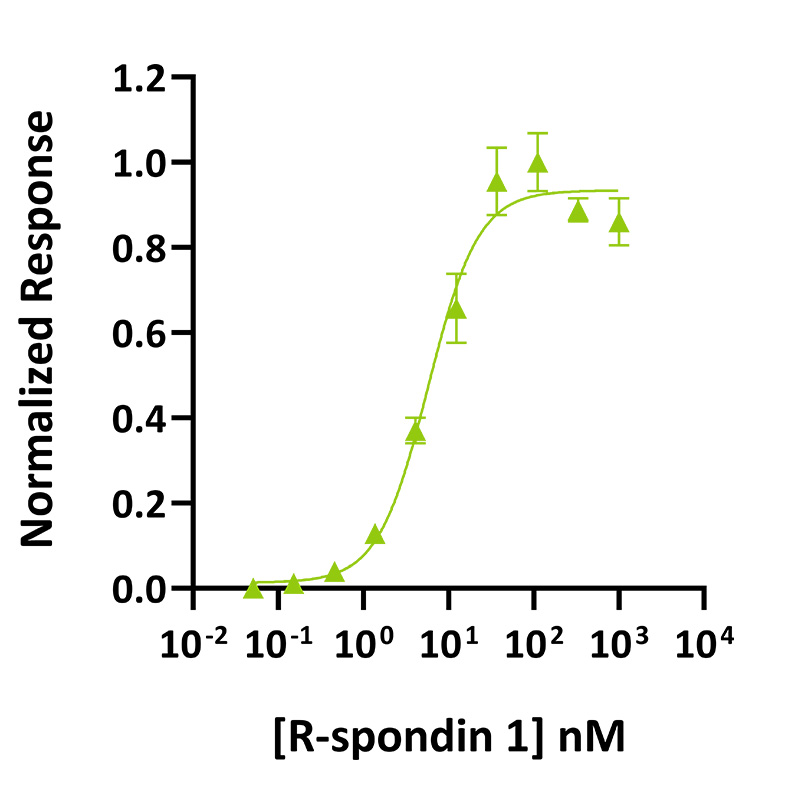
R-spondin 1 (Qk006) activity was determined using the Wnt-responsive firefly luciferase reporter assay as it enhances Wnt-ß catenin signaling in HEK239T cells. HEK293T cells transfected with reporter TOP-FLASH are treated in triplicate with increasing concentration of R-spondin 1 (diluted in DMEM with 0.5 % of FCS), in the presence of Wnt-conditioned media (1:8 dilution). Cells are grown overnight, and luciferase activity is measured and normalized. EC50 = 5.8 nM (75.1 ng/mL). Data from Qk006 lot #104277.
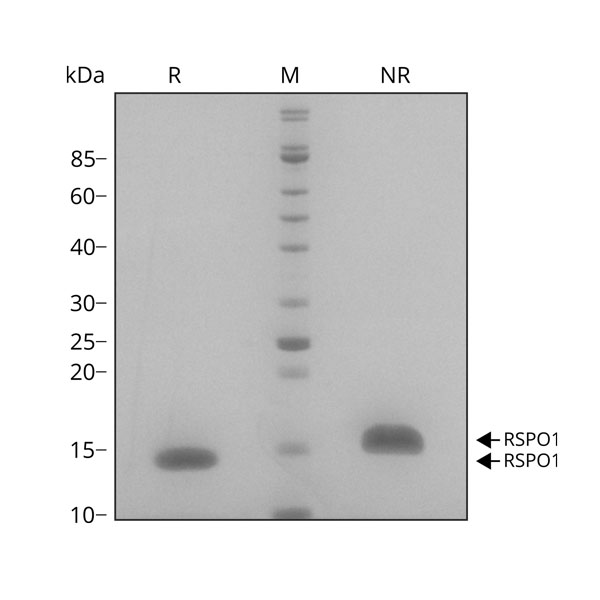
R-spondin 1 migrates as a single band at 16 kDa in non-reducing (NR) and 13 kDa in reducing (R) conditions. Purified recombinant protein (7 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptothanol, R) and non-reduced conditions (NR) and stained with Coomassie Brilliant Blue R250. Data from Qk006 lot #010.
Mass spectrometry: single species with expected mass
Recovery from stock vial: >95%
Endotoxin: <0.05 EU/μg protein
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.

Qkine R-spondin 1 bioactivity was comparable to an alternative supplier Rspondin-1. Bioactivity was determined using the Wnt-responsive firefly luciferase reporter assay as it enhances Wnt-ß catenin signaling in HEK239T cells. HEK293T cells transfected with reporter TOP-FLASH are treated in triplicate with increasing concentration of Qkine R-spondin 1 (Qk006, green) or alternative R-spondin 1 (Supplier B, black), in the presence of Wnt-conditioned media (1:8 dilution). Cells are grown overnight, and luciferase activity is measured and normalized.
R-spondin 1 (RSPO-1) is a secreted protein of the RSPO family that regulates the canonical Wnt signaling pathway [1,2]. The Wnt pathway is a highly conserved signaling pathway that is essential for various cellular processes, including cell proliferation, differentiation, and migration. This growth factor plays a crucial role in embryonic development and patterning, particularly in the development of the gastrointestinal tract, neural tube, skin, and limbs [3]. It is involved in the maintenance of stem cells and can enhance the self-renewal capacity of stem cells, promoting tissue regeneration and repair [4]. It is also involved in the sex determination and the development and maintenance of the female sex characteristics in particular the differentiation and maintenance of the ovarian tissues [5].
RSPO1 proteins are generally characterized by a core domain known as the thrombospondin type 1 (TSP1) domain, which is responsible for their signaling activity. This protein is comprised of 263 amino acids and structured as two cysteine-rich, furin-like domains [3,6]. It acts as an agonist (activator) of the Wnt signaling pathway by binding to the leucine-rich repeat-containing G-protein coupled receptors (LGR) 4-6, leading to the stabilization of β-catenin, a key component of the pathway [7].
In cell culture, this recombinant protein is used extensively for the maintenance of self-renewal and expansion of stem cells. It is also used for the culture of intestinal, hematopoietic, and epithelial stem cells [2,7,8]. Also, the culture of stem cell-derived organoids such as intestinal, pancreas liver, colon, and stomach organoids benefit from the inclusion of RSPO1 in the media [7,9,10].
Dysregulation of the Wnt signaling pathway, including the overexpression of R-spondin 1, has been associated with tumor growth and progression in various cancers such as colon and ovarian cancer [11,12]. Research is ongoing on the role of R-spondin 1 in various biological processes and its potential as a therapeutic target for cancer treatment. Given its role in stem cell maintenance and tissue regeneration, RSPO1 and related proteins have also been explored as potential therapeutic targets for regenerative medicine.
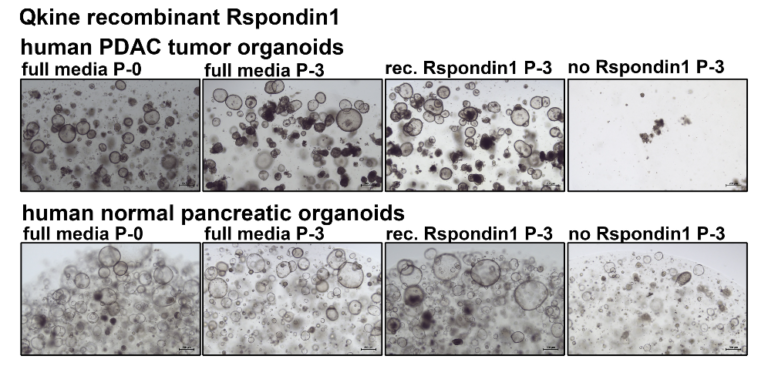
Comparison between human healthy pancreatic organoids and human and pancreatic ductal adenocarcinoma (PDAC) tumor organoids. Initial cultures were maintained in full growth media supplemented with Wnt3a R-spondin 1 conditioned media (full media P-0). Cultures were continued for 3 passages with Wnt3a R-spondin 1 conditioned media (full media P-3), growth media with 100 nM recombinant R-spondin 1 (Rec. R-spondin 1), or growth media alone (No R-spondin 1). No differences in organoid growth were observed when using recombinant R-spondin 1 instead of R-spondin 1 conditioned media, whereas removing R-spondin 1 prevented organoid formation.
Experiments have been conducted by Dennis Plenker, Ph.D. in the lab of Dr David Tuveson at Cold Spring Harbor Laboratory.
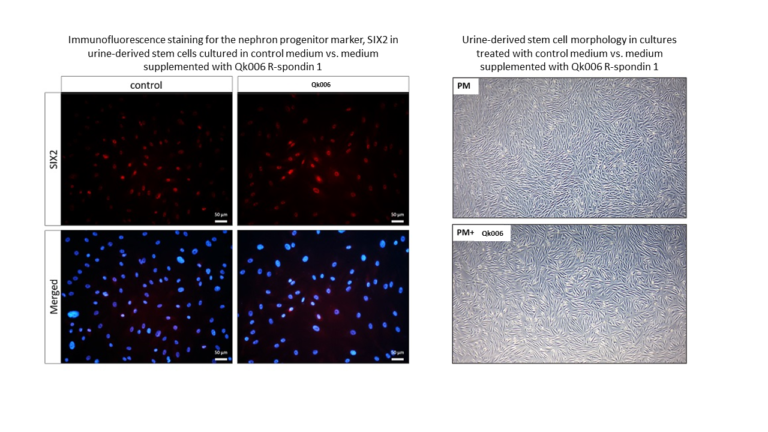
Qkine R-spondin 1 treatment of urine-derived stem cells maintains cellular morphology and nuclear expression of the nephron progenitor marker, SIX2.
Previous research suggests that the removal of R-spondin 1 from the culture media of urine-derived stem cells leads to loss of kidney progenitor molecules in mice. Therefore, it was hypothesized that the addition of R-spondin 1 to the culture media would result in maintenance of SIX2 progenitor molecules and thus maintenance of the undifferentiated state.
Urine-derived stem cells were treated with R-spondin 1 (Qk006) at a concentration of 200 ng/ml for 5 days. The cells were then fixed and stained for SIX2.
Recombinant human R-spondin 1 protein maintains urine-derived stem cell morphology. Urine-derived stem cells typically have a small, rice grain-like morphology and express nuclear SIX2. Comparing the morphology of untreated vs treated cells showed that cells treated with R-spondin 1 (Qk006) had a similar morphology to the control, indicating that R-spondin 1 treatment maintains the undifferentiated state.
Recombinant human R-spondin 1 protein maintains nuclear SIX2 expression in urine-derived stem cells. In urine-derived stem cells, SIX2 expression is predominately found in the cell nuclei, as shown in the untreated control. Cytoplasmatic expression of SIX2 was also observed, which indicates cell differentiation. Addition of R-spondin 1 (Qk006) maintained nuclear SIX2 expression (as shown by immunofluorescence staining for SIX2) and therefore appears to keep the urine-derived stem cells in an undifferentiated state.
Experiments have been conducted by Lisa Nguyen (MSc) and team in the laboratory of Professor James Adjaye at the Institute for Stem Cell Research and Regenerative Medicine, University of Dusseldorf.
Choi W, Kim YH, Woo SM et al.
DOI: doi: 10.4143/crt.2022.1630Agarwal R, Dittmar T, Beer HD et al.
DOI: doi: 10.1111/exd.14823Osorio-Vasquez V, Lumibao JC, Peck KL et al.
DOI: https://doi.org/10.1101/2024.10.30.620903Ku B, Eisenbarth D, Baek S et al.
DOI: doi: 10.1016/j.xcrm.2024.101461Lumibao JC, Okhovat SR, Peck KL et al.
DOI: doi: 10.1172/jci.insight.172419Lee, Mi Rim et al.
DOI: doi: 10.1016/j.xcrm.2025.102129Kirino S, Uefune F, Miyake K et al.
DOI: doi: 10.1038/s42003-026-09533-xKang S, Lee MR, Choi W, Kong S-Y and Kim Y-H
DOI: doi.org/10.1016/j.xpro.2025.104039Sung Y, Yu YC, Lee M et al.
DOI: doi.org/10.1038/s41467-025-64730-2Yamagchi A, Ishikawa K and Akamatsu W
DOI: doi.org/10.64898/2026.02.07.704302Tsang CF, Patel H, Kouassi FM et al.
DOI: DOI: 10.1002/path.70025Uefune F and Yui S
DOI: https://doi.org/10.1016/j.bbrc.2026.153645Park M-S, Gunassekaran GR, Lee S-M et al.
DOI: DOI: 10.34133/bmr.0361R-spondin 1 regulates Wnt signaling and β-Catenin stabilization. Therefore, the functions of R-spondin 1 are diverse and are associated with cell proliferation and differentiation. It maintains the self-renewal capacity of stem cells and is involved in the embryonic development especially the development of the female reproductive system, gastrointestinal track, and neural tube.
The Wnt signaling pathway is involved in in various biological processes. It is involved in the cell fate specification, embryonic development, stem cell maintenance, tissue regeneration, synaptic function, immune response, cancer progression, and cell migration.
The molecular weight is 13 kDa.
The RSPO1 gene provides instructions for producing the R-spondin 1 protein.
The RSPO1 gene is located at 1q41.2.
It is a growth factor supplemented to cell culture media. It is used for the culture of induced pluripotent stem cells, embryonic stem cells, epithelial stem cells, and organoids such as intestinal and colon organoids.
The size of the R-spondin proteins, including R-spondin 1 (RSPO1), can vary slightly depending on the specific isoform and the species being considered. In humans, RSPO1 is approximatively 263 amino acids in length with a molecular weight of 13-29 kDa.
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.
For use in manufacturing of cellular or gene therapy products. Not intended for in vivo applications.

£100, $140 or €120 Qkine gift card for product reviews with an image and £50, $70 or €60 for reviews without an image.

