

Price range: £155.00 through £1,540.00
Recombinant porcine HGF protein is a potent, high-purity NK1 isoform of porcine hepatocyte growth factor (HGF). The porcine recombinant protein HGF differs from human HGF at several key amino acid residues that are predicted to affect bioactivity and receptor binding. Use of species-specific HGF growth factor will facilitate media optimization for cellular agriculture (cultivated meat) and veterinary applications. Also available is Qk060 bovine HGF NK1. The highly scalable animal origin-free manufacture and enhanced bioactivity make this suitable for chemically-defined media and reproducible scale-up.
Qk061 is a 20 kDa naturally occurring isoform of HGF, animal origin-free and carrier-protein free.
In stock
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
Price range: £155.00 through £1,540.00
Buy online with secure credit card or purchase order. For any questions, please email orders@qkine.com
>98%, by SDS-PAGE quantitative densitometry
Expressed in E. coli
Animal origin-free (AOF) and carrier protein-free
Manufactured in our Cambridge, UK laboratories
Lyophilized from acetonitrile, TFA
Differentiation of iPSCs to hepatocyte-like cells
iPSC and primary cell differentiation to muscle
Cellular agriculture and cultivated meat cell culture media optimization
Cellular agriculture process development

HGF NK1 activity was determined using the Promega serum response element luciferase reporter assay (*) in HEK293T cells. EC50 = 7.0 ng/ml (338 pM). Cells were treated in triplicate with a serial dilution of HGF NK1 for 3 hours. Firefly luciferase activity was measured and normalized to the control Renilla luciferase activity. Data from Qk061 lot #104417. Please note: protein activity has been determined in a standardized assay using human cells. *Promega pGL4.33[luc2P/SRE/Hygro] #E1340
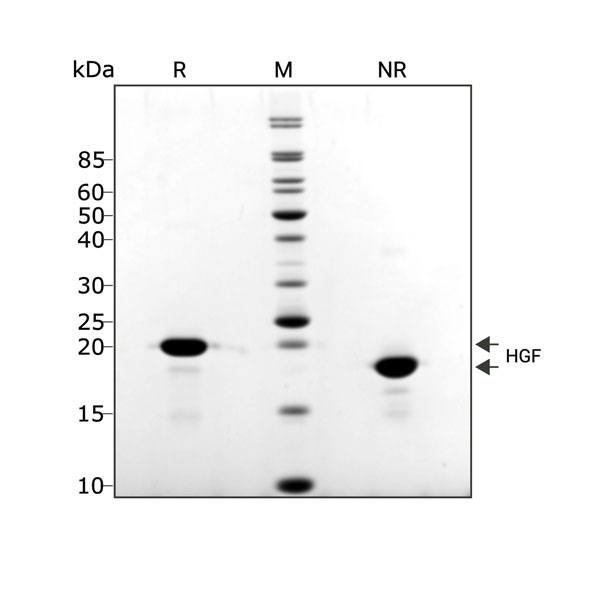
Recombinant porcine HGF protein (Qk061) migrates as a single band at 18 kDa in non-reducing (NR) conditions and 20 kDa upon reduction (R). Purified recombinant protein (3 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptothanol, R) and non-reduced (NR) conditions and stained with Coomassie Brilliant Blue R250. Data from Qk061 batch #104417.
Mass spectrometry: single species with expected mass
Recovery from stock vial: >95%
Endotoxin: <0.05 EU/μg protein
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.

Species-specificity of HGF is predicted to impact bioactivity. Porcine HGF differs from human and bovine HGF at several key amino acid residues that are predicted to affect bioactivity and receptor binding. Clustal Alignment showing sequence variation between HGF NK1 from different species. Note the lack of structural conservation at key residues, which is predicted to impact receptor binding and activation.
Hepatocyte growth factor (HGF) regulates cell growth, cell motility, and morphogenesis and plays a central role in angiogenesis, tumorogenesis, and tissue regeneration. HGF binds and activates the receptor tyrosine kinase, c-Met, activating PI3K/AKT, FAK, JNK, and ERK1/2 signaling [1].
HGF is secreted as a single inactive polypeptide and is cleaved by serine proteases into a 62-kDa heavy-chain and 32-36-kDa light-chain. A disulfide bond between the heavy and light chains produces the active, heterodimeric molecule [2]. Alternative splicing of HGF produces multiple transcript variants encoding different isoforms. The NK1 isoform is the smallest naturally occurring splice variant, comprising the N-terminus and first kringle domain [3].
Bovine, porcine and human HGF are all members of the hepatocyte growth factor (HGF) family. While the sequence of porcine HGF is similar to human HGF, there are differences in structure and bioactivity. Porcine HGF is more resistant to protease digestion. Additionally, porcine HGF appears to be more potent than its counterparts when it comes to inducing muscle cell proliferation and differentiation. These findings demonstrate that porcine HGF could have important implications for cellular agriculture applications as it may provide a faster route towards creating tissues with improved regenerative qualities.
Qkine HGF (NK1) recombinant protein is produced in an animal-free microbial system that is compatible with production at scale. We are expanding our range of innovative growth factors for cellular agriculture and cultivated meat media development to support advances in the field, please reach out to cellag@qkine.com to discuss collaboration opportunities.
HGF stands for hepatocyte growth factor, a multifunctional cytokine.
HGF is a protein that is found in various tissues and organs throughout the body such as the liver, kidneys, lungs, pancreas, blood vessels, muscles, central nervous system, and placenta.
HGF is produced by various cell types including mesenchymal cells, hepatocytes, smooth muscle cells, epithelial cells, firbroblasts, and other stromal cells.
The alternative name of HGF is scatter factor (SF).
Yes, it is a multifunctional cytokine.
The HGF gene, also know as SF, HGFB, HPTA, F-TCF or DFNB39 provides instructions for synthesizing the protein HGF. Alternative splicing results in multiple transcript variants, at least one of which encodes a preproprotein that is proteolytically processed to generate alpha and beta chains, which form the mature heterodimer.
HGF binds to Hepatocyte Growth Factor Receptor (HGFR), also known as c-Met.
The Hepatocyte Growth Factor Receptor (HGFR), also known as c-Met, is a receptor tyrosine kinase that plays a crucial role in mediating the biological effects of Hepatocyte Growth Factor (HGF). The c-Met receptor is expressed on the surface of various cell types, and its activation initiates a cascade of intracellular pathways involved in cell proliferation, migration, angiogenesis, regeneration, and embryonic development.
HGF pathway, also known as the HGF/c-Met pathway, involves a series of molecular events triggered by the binding of HGF to its receptor, c-Met. It activates downstream signalling pathways such as the RAS-RAF-MAPK, PI3K-AKT, STAT3, and Wnt/β-catenin Pathways.
In cell culture, HGF is used for various applications such as the differentiation of specific lineages in particular mesenchymal cells and the growth, survival, and maintenance of hepatocytes and endothelial cells. HGF is also used in assays to study cell cycle, division, motility, invasion, and wound healing.
Yes, HGF regulates the differentiation of mesenchymal cells into hepatocytes, osteocytes and endothelial cells.
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.
For use in manufacturing of cellular or gene therapy products. Not intended for in vivo applications.

£100, $140 or €120 Qkine gift card for product reviews with an image and £50, $70 or €60 for reviews without an image.

