 Recombinant FGF2-G3 (145 aa) protein (Qk052)
Recombinant FGF2-G3 (145 aa) protein (Qk052)Recombinant FGF2-G3 (145 aa) protein (Qk052)
Price range: £160.00 through £800.00
FGF2-G3 (145 aa) protein is a thermostable engineered form of FGF-2 (bFGF). Qk052 comprises the 145 aa form of FGF-2 (Qk025) with the nine amino acid substitutions developed by Dvorak et al. 2018. This increases the functional half-life of the protein from <10 h (wild-type) to >7 days (FGF2-G3 145 aa).
FGF2-G3 is used in B8 media (Kuo et al. 2019) for weekend free, high homogeneity induced pluripotent stem cell culture. This new 145 aa thermostable FGF-2 protein allows direct comparison of efficacy in systems using FGF-2 145 aa (Qk025) for chemically defined stem cell and organoid culture media, and cultured meat media development. High purity 16 kDa bioactive FGF2-G3 145 aa protein, animal origin-free (AOF), carrier protein-free and with no His-tag.
Product manufactured under license: International Patent Application No. PCT/EP2016/073567 and U.S. Patent No. 11,746,135.
In stock
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
Price range: £160.00 through £800.00
Fast and free shipping.
Buy online with secure credit card or purchase order. For any questions, please email orders@qkine.com
Summary:
- High purity thermostable FGF2-G3 protein comprising 145 aa form of FGF-2 (Uniprot: P09038) with nine stabilizing amino acid substitutions
- 16 kDa
>98%, by SDS-PAGE quantitative densitometry
Expressed in E. coli
Animal origin-free (AOF) and carrier protein-free
Manufactured in our Cambridge, UK laboratories
Lyophilized from Tris, NaCl, CyS, mannitol
- Resuspend in sterile-filtered water at >50 µg/ml, add carrier protein if desired, prepare single use aliquots and store frozen at -20 °C (short-term) or -80 °C (long-term).
Featured applications:
Induced pluripotent stem cell culture and maintenance
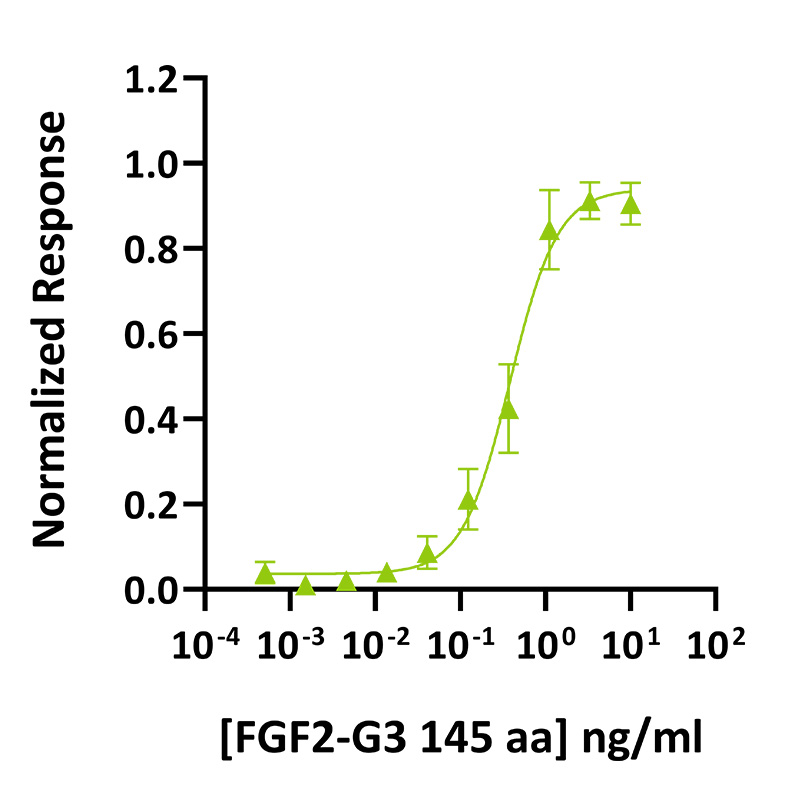
FGF2-G3 (145 aa) activity was determined using the Promega serum response element luciferase reporter assay in transfected HEK293T cells. Cells were treated in triplicate with a serial dilution of FGF2-G3 (145 aa) for 3 hours. Firefly luciferase activity was measured and normalized to the control Renilla luciferase activity. Data from Qk052 lot #204567. EC50 = 0.38 ng/ml.
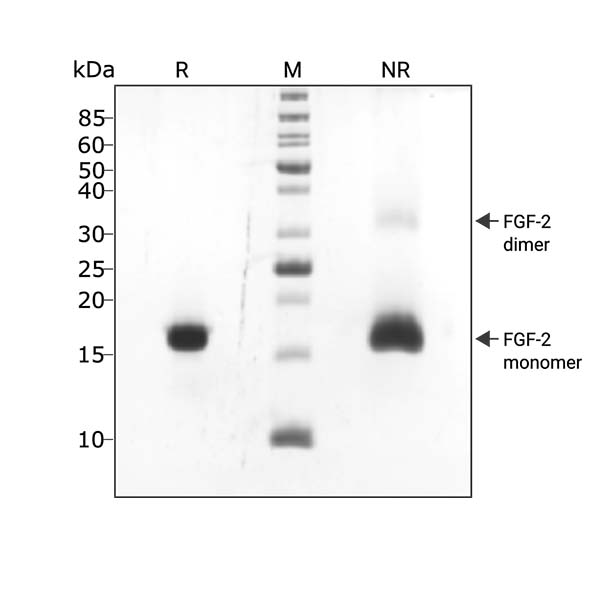
FGF2-G3 (145 aa) migrates as a major band at 16 kDa in non-reducing (NR) conditions. A small amount of dimer can be seen (higher molecular weight band). Upon reduction (R), only the 16 kDa band is visible. No contaminating protein bands are present. Purified recombinant protein (3 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptothanol, R) and non-reduced (NR) conditions and stained with Coomassie Brilliant Blue R250. Data from Qk052 batch #104339.
Further quality assays
Mass spectrometry: single species with expected mass
Recovery from stock vial: >95%
Endotoxin: <0.005 EU/μg protein (below level of detection)
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.

WT FGF2 (Qk025) and FGF2-G3 (Qk052) were diluted in conditioned media and incubated at 37oC for 2 or 7 days before being serially diluted and then assayed in triplicate for activity using the Promega serum response element luciferase reporter assay in transfected HEK293T cells. Firefly luciferase activity was measured and normalized to the control Renilla luciferase activity.
Protein background
FGF-2 (also known as basic FGF or bFGF) is an essential growth factor for maintaining human embryonic stem cell (hESC) and induced pluripotency stem cell (iPSC) pluripotency in feeder-free and chemically defined stem cell media. It is a core component of widely adopted media including mTESR [1, 2], StemPRO [3] and E8 [4]. However, FGF-2 is inherently unstable and prone to proteolytic degradation and aggregation. This fundamental biochemical instability, and therefore low half-life in culture media (<10 h), is an important contribution to the need for frequent media changes and challenges in improving homogeneity during stem cell proliferation and subsequent differentiation.
To improve the stability of FGF-2 for stem cell media and regenerative medicine applications, Dvorak and colleagues at Masaryk University used computer-assisted protein engineering to identify an optimal set of nine amino acid substitutions that stabilize FGF-2. These substitutions were designed to avoid structural changes to the FGF receptor 1 (FGFR1) and FGF receptor 2 (FGFR2) binding sites. This thermostable FGF-2 is known as FGF2-G3, or FGF2-STAB® [5]. The biological activity of wild-type FGF-2 is <50% after 10h incubation with conditioned media. In contrast, no reduction in FGF2-G3 biological activity is observed after >7 days incubation with conditioned media at 37oC. Both FGF2-G3 and wild-type FGF-2 maintain hESC pluripotency and expression of pluripotency markers Oct-4 and nanog with equivalent efficacy [5].
In 2020, Paul Burridge and colleagues at Northwestern University, Chicago, published a protocol for B8 media. This iPSC maintenance media uses thermostable FGF2-G3, along with optimization of media component concentration and composition to reduce media cost and facilitate weekend-free stem cell culture regimes [6, 7].
To manufacture and provide FGF2-G3 for stem cell culture, including use in B8 media and emerging applications such as cultured meat, Qkine has licensed the patented FGF2-G3 technology from Enantis/Masaryk University. We have combined the excellent science behind the FGF2-G3 technology with our protein manufacture expertise to provide gold standard protein for use in cell culture media. We have removed His tags present in academic forms of the protein, as these may give rise to issues for scientists translating discoveries to the clinical or scale-up. His tags also introduce unnecessary scientific uncertainty.
Additional resources
- Differentiation of induced pluripotent stem cells (iPSCs) into neuroectoderm (PDF)
- Differentiation of induced pluripotent stem cells (iPSCs) into mesoderm (PDF)
- CytoBoost™ for increased proliferation of muscle and fat cells in serum-free cultures
- Brochure: FGF2-G3 Thermostable FGF-2 for enhanced stem cell culture
- Brochure: Growth factors for neural and glial cell differentiation
- Brochure: Growth factors for enhanced organoid culture protocols
Publications using Recombinant FGF2-G3 (145 aa) protein (Qk052)
-
Refined home-brew media for cost-effective, weekend-free hiPSC culture and genetic engineering
Truszkowski L, Bottini S, Bianchi S et al.
DOI: https://doi.org/10.12688/openreseurope.18245.1
FAQ
Fibroblast growth factor 2 (FGF-2), also known as basic fibroblast growth factor (bFGF) is a growth factor and signaling protein.
FGF-2 is expressed in a developmental and tissue specific manner. It’s expression is tightly controlled in normal tissues and it can be detected in all major tissues.
FGF-2 is essential for normal embryonic development. It has roles in cell survival and proliferation, angiogenesis, tumorigenesis, wound healing and tissue repair.
FGF-2 binds to and signals though all four of the FGF receptors FGFR1-4.
FGFRs phosphorylate specific tyrosine residues and activate the RAS-MAPK, PI3K-AKT, PLCγ, and STAT intracellular signaling pathways.
FGF-2 is used to maintain the pluripotency of stem cells in culture.
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.

Receive an Amazon gift voucher when you leave us a review.
£25, $30 or €30 for reviews with an image and £10, $15 or €15 for reviews without an image
