Currency
Recombinant human HGF (NK1) protein (Qk013)
Hepatocyte growth factor (HGF) is a multi-functional cytokine that promotes the growth and proliferation of various cell types including mesenchymal, endothelial, and epithelial cells. It is involved in embryonic development, cell motility, survival, angiogenesis, and tissue regeneration. In cell culture, HGF is used to promote the differentiation of mesenchymal cells towards hepatocytes, osteocytes, and endothelial cells.
Recombinant human HFG NK1 protein is a potent naturally occurring isoform of HGF. This protein is animal-free, carrier protein-free, and tag-free to ensure its purity with exceptional lot-to-lot consistency with a molecular weight of 20 kDa. It promotes efficient differentiation of human induced pluripotent stem cells to hepatocyte-like cells at just 10 ng/ml with highly homogeneous expression of the hepatic marker, HNF4α.
Orders are typically shipped same or next day (except Friday).
Easy world-wide ordering, direct or through our distributors.
1000µg will be despatched as 2 x 500µg
Buy online with secure credit card or purchase order or email orders@qkine.com
Bulk and stock reservation available
Summary
High purity human HGF (NK1) protein (Uniprot: P14210)
>98%, by SDS-PAGE quantitative densitometry
20 kDa
Expressed in E. coli
Animal-free (AOF) and carrier protein-free.
Manufactured in our Cambridge, UK laboratories
Lyophilized from acetonitrile, TFA
Resuspend in 10mM HCl at >100 µg/ml (provided with protein and free of charge), prepare single use aliquots, add carrier protein if desired and store frozen at -20°C or -80°C
Featured applications
Differentiation of human iPSCs to hepatocyte-like cells at just 10 ng/ml
[1] M. C. Stella and P. M. Comoglio, “HGF: a multifunctional growth factor controlling cell scattering,” Int. J. Biochem. Cell Biol., vol. 31, no. 12, pp. 1357–1362, Dec. 1999, doi: 10.1016/S1357-2725(99)00089-8.
[2] T. Nakamura and S. Mizuno, “The discovery of Hepatocyte Growth Factor (HGF) and its significance for cell biology, life sciences and clinical medicine,” Proc. Jpn. Acad. Ser. B, vol. 86, no. 6, pp. 588–610, 2010, doi: 10.2183/pjab.86.588.
[3] B. Niranjan et al., “HGF/SF: a potent cytokine for mammary growth, morphogenesis and development,” Development, vol. 121, no. 9, pp. 2897–2908, Sep. 1995, doi: 10.1242/dev.121.9.2897.
[4] H. Ohmichi, U. Koshimizu, K. Matsumoto, and T. Nakamura, “Hepatocyte growth factor (HGF) acts as a mesenchyme-derived morphogenic factor during fetal lung development,” Development, vol. 125, no. 7, pp. 1315–1324, Apr. 1998, doi: 10.1242/dev.125.7.1315.
[5] G. Forte et al., “Hepatocyte Growth Factor Effects on Mesenchymal Stem Cells: Proliferation, Migration, and Differentiation,” Stem Cells, vol. 24, no. 1, pp. 23–33, Jan. 2006, doi: 10.1634/stemcells.2004-0176.
[6] B. Zheng et al., “Neural differentiation of mesenchymal stem cells influences chemotactic responses to HGF,” J. Cell. Physiol., vol. 228, no. 1, pp. 149–162, 2013, doi: 10.1002/jcp.24114.
[7] P. P. Wang et al., “Expression of hepatocyte-like phenotypes in bone marrow stromal cells after HGF induction,” Biochem. Biophys. Res. Commun., vol. 320, no. 3, pp. 712–716, Jul. 2004, doi: 10.1016/j.bbrc.2004.05.213.
[8] S.-H. Oh et al., “Hepatocyte Growth Factor Induces Differentiation of Adult Rat Bone Marrow Cells into a Hepatocyte Lineage in Vitro,” Biochem. Biophys. Res. Commun., vol. 279, no. 2, pp. 500–504, Dec. 2000, doi: 10.1006/bbrc.2000.3985.
[9] S. L. Organ and M.-S. Tsao, “An overview of the c-MET signaling pathway,” Ther. Adv. Med. Oncol., vol. 3, no. 1 Suppl, pp. S7–S19, Nov. 2011, doi: 10.1177/1758834011422556.
[10] A. G. Oliveira, T. G. Araújo, B. de M. Carvalho, G. Z. Rocha, A. Santos, and M. J. A. Saad, “The Role of Hepatocyte Growth Factor (HGF) in Insulin Resistance and Diabetes,” Front. Endocrinol., vol. 9, 2018, Accessed: Dec. 18, 2023. [Online]. Available: https://www.frontiersin.org/articles/10.3389/fendo.2018.00503
[11] J. L. Jakubczak, W. J. Larochelle, and G. Merlino, “NK1, a Natural Splice Variant of Hepatocyte Growth Factor/Scatter Factor, Is a Partial Agonist In Vivo,” Mol. Cell. Biol., vol. 18, no. 3, pp. 1275–1283, Mar. 1998, doi: 10.1128/MCB.18.3.1275.
[12] F. Cecchi, D. C. Rabe, and D. P. Bottaro, “Targeting the HGF/Met signalling pathway in cancer,” Eur. J. Cancer, vol. 46, no. 7, pp. 1260–1270, May 2010, doi: 10.1016/j.ejca.2010.02.028.
[13] F. Cecchi, D. C. Rabe, and D. P. Bottaro, “Targeting the HGF/Met signaling pathway in cancer therapy,” Expert Opin. Ther. Targets, vol. 16, no. 6, pp. 553–572, Jun. 2012, doi: 10.1517/14728222.2012.680957.
Human
Species similarity:
Porcine – 95%
Bovine – 93%
Rat – 90%
Mouse – 89%
Frequently used together
Bioactivity
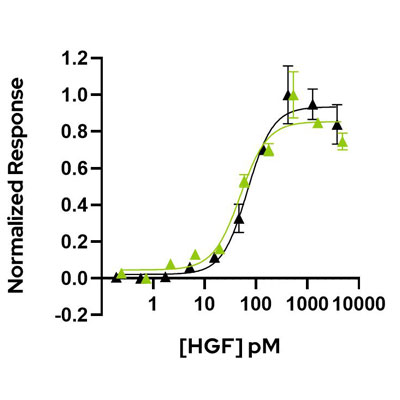
HGF (NK1) (Qk013) has equivalent bioactivity to a mammalian cell-expressed full-length HGF from another commercial supplier. Comparative bioactivity was determined using a quantitative Promega serum response element luciferase reporter assay in transfected HEK293T cells. Cells were treated (in duplicate) with a serial dilution of HGF for 6 hours. Firefly luciferase activity is measured and normalized to control Renilla luciferase activity.
Qk013 HGF (green) has an EC50 of 49.6 pM (0.99 ng/mL) and the mammalian cell-expressed HGF (black) has an EC50 of 68.4 pM (1.37 ng/mL).
Data from Qk013 lot #010.
Purity
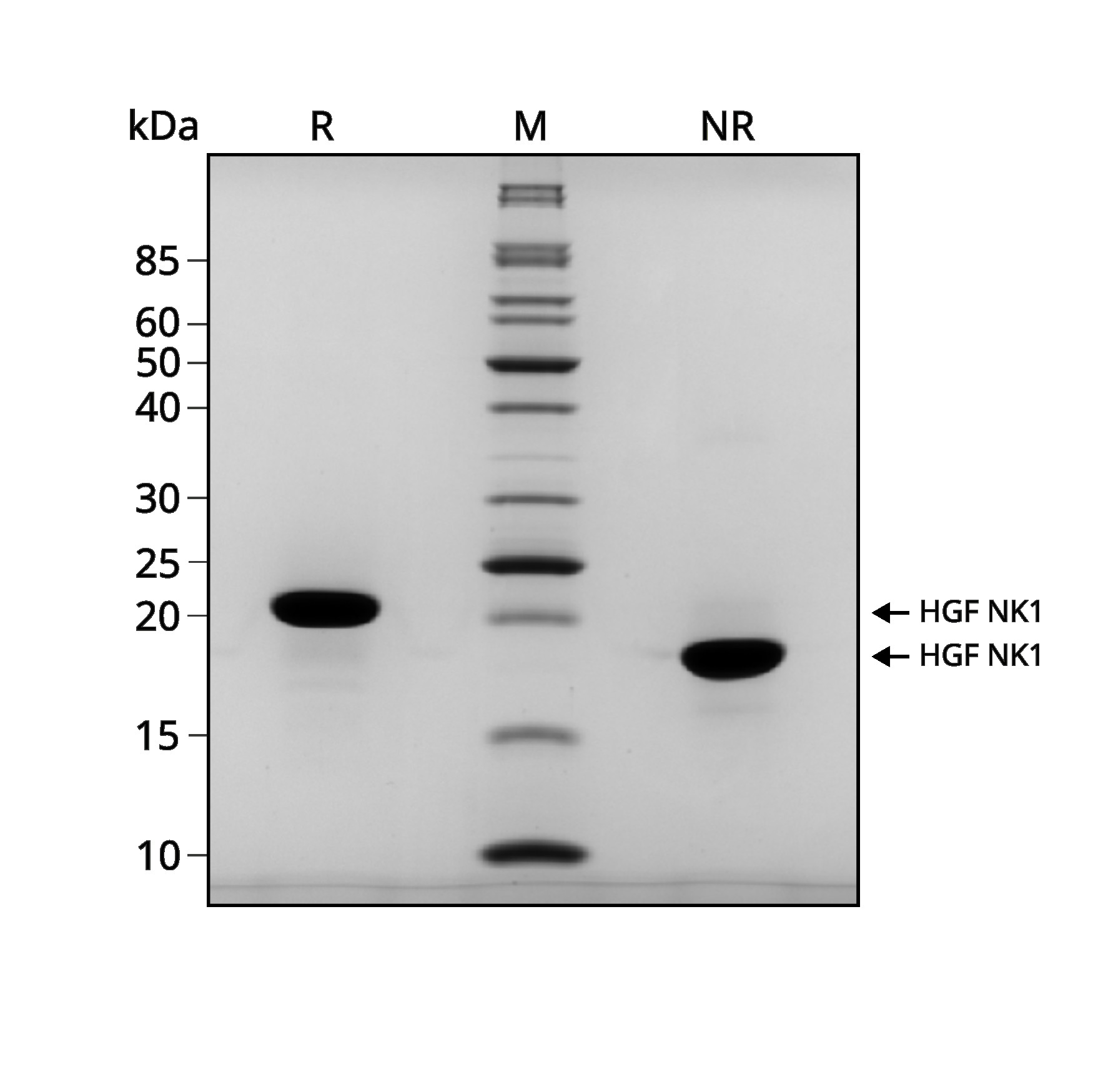
Recombinant HGF NK1 migrates as a major band at approximately 21 kDa in reduced (R) and at approximately 18 kDa in non-reduced (NR) conditions. No contaminating protein bands are present.
The purified recombinant protein (3 µg) was resolved using 15% w/v SDS-PAGE in reduced (+β-mercaptoethanol, R) and non-reduced (NR) conditions and stained with Coomassie Brilliant Blue R250.
Data from Qk013 lot #104451.
Further quality assays
Mass spectrometry: single species with expected mass
Analytical reversed-phase: single sharp peak
Endotoxin: <0.005 EU/μg protein (below level of detection)
Recovery from stock vial: >95%
We are a company founded and run by scientists to provide a service and support innovation in stem cell biology and regenerative medicine. All our products are exceptionally high purity, with complete characterisation and bioactivity analysis on every lot.
Protein background
Hepatocyte growth factor (HGF), or scatter factor (SF), is a multifunctional cytokine that acts as a mitogen and promotes the growth and proliferation of various cell types, including mesenchymal, endothelial, and epithelial cells [1], [2]. During embryonic development, it stimulates the development of various organs, such as the liver, kidneys, and lungs [2]–[4]. It plays a crucial role in adulthood by regulating cell motility, survival, angiogenesis, and tissue regeneration, particularly in liver regeneration [2]. It is also involved in tissue homeostasis and is released triggered by factors such as injury, stress, and inflammation. HGF has been implicated in promoting osteogenic, chondrogenic, and adipogenic differentiation of mesenchymal cells [3], [4].
In cell culture, HGF is used for various applications. HGF modulates cell fate and promotes the differentiation of specific lineages in particular mesenchymal cells [5], [6]. HGF is essential for the growth, survival, and maintenance of hepatocytes and endothelial cells [7], [8]. Finally, HGF is used to study cell cycle, division, motility, invasion, and wound healing [7].
HGF binds and activates the receptor tyrosine kinase, c-Met; through this receptor, HGF activates PI3K/AKT, FAK, JNK, and ERK1/2 signaling [1], [9]. HGF is secreted as a single inactive polypeptide and is cleaved by serine proteases into a 62-kDa heavy-chain and 32-36-kDa light-chain [1]. A disulfide bond between the heavy and light chains produces the active αβ heterodimeric molecule [1], [10]. Alternative splicing of HGF produces multiple transcript variants encoding different isoforms. The NK1 isoform is the most minor naturally occurring splice variant, it comprises the HGF N-terminal hairpin loop domain (involved in the c-Met receptor binding) and the first kringle domain [11].
Due to its involvement in cell growth and tissue repair, HGF has attracted attention in regenerative medicine and potential therapeutic interventions. Additionally, the dysregulation of HGF and its receptor c-Met has been implicated in various diseases, including cancer, where aberrant signaling contributes to tumor growth, invasion, and metastasis [1], [2], [12], [13].
Customer & collaborator data
High purity, animal and carrier protein-free HGF protein for chemically-defined media and more reproducible scale-up
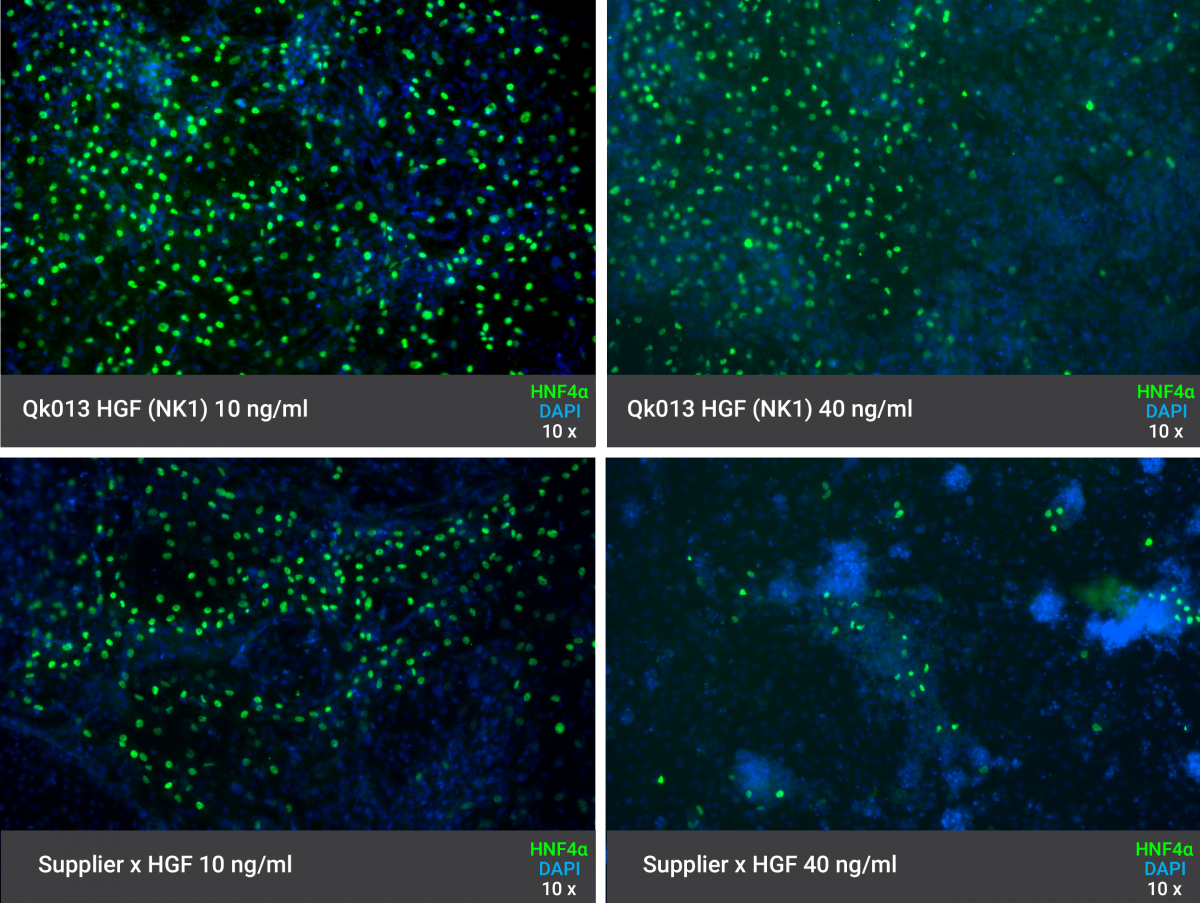
Recombinant human HGF (NK1) protein promotes efficient differentiation of human iPSCs to hepatocyte-like cells
HGF (NK1) supports human iPSC differentiation to hepatocyte-like cells. Defined media was supplemented with Qk013 HGF (NK1) during day 8-18 of the hepatocyte differentiation protocol (adapted from Heck W, et al. 2015). Cells attain characteristic hexagonal epithelial morphology. Expression of hepatic marker, HNF4α, and polarisation marker, E-Cadherin (ECAD), was detected using immunofluorescence.
Highly homogeneous expression of HNF4α in hepatocyte-like cells differentiated from iPSCs using Qk013 HGF (NK1)
Growth in media supplemented with Qk013 HGF (NK1) leads to highly homogeneous expression of hepatic marker, HNF4α. iPSCs were treated with hepatocyte differentiation media supplemented with Qk013 HGF (NK1) or full-length, mammalian cell-derived HGF from another commercial source.
All experiments have been conducted by the specialist stem cell biotechnology company, Stemnovate Limited, in Cambridge, UK.
FAQ
HGF stands for hepatocyte growth factor, a multifunctional cytokine.
HGF is a protein that is found in various tissues and organs throughout the body such as the liver, kidneys, lungs, pancreas, blood vessels, muscles, central nervous system, and placenta.
HGF is produced by various cell types including mesenchymal cells, hepatocytes, smooth muscle cells, epithelial cells, firbroblasts, and other stromal cells.
The alternative name of HGF is scatter factor (SF).
Yes, it is a multifunctional cytokine.
The HGF gene provides instructions for synthesizing the protein HGF.
HGF binds to Hepatocyte Growth Factor Receptor (HGFR), also known as c-Met.
The Hepatocyte Growth Factor Receptor (HGFR), also known as c-Met, is a receptor tyrosine kinase that plays a crucial role in mediating the biological effects of Hepatocyte Growth Factor (HGF). The c-Met receptor is expressed on the surface of various cell types, and its activation initiates a cascade of intracellular pathways involved in cell proliferation, migration, angiogenesis, regeneration, and embryonic development.
HGF pathway, also known as the HGF/c-Met pathway, involves a series of molecular events triggered by the binding of HGF to its receptor, c-Met. It activates downstream signalling pathways such as the RAS-RAF-MAPK, PI3K-AKT, STAT3, and Wnt/β-catenin Pathways.
In cell culture, HGF is used for various applications such as the differentiation of specific lineages in particular mesenchymal cells and the growth, survival, and maintenance of hepatocytes and endothelial cells. HGF is also used in assays to study cell cycle, division, motility, invasion, and wound healing.
Yes, HGF regulates the differentiation of mesenchymal cells into hepatocytes, osteocytes, endothelial cells…
Our products are for research use only and not for diagnostic or therapeutic use. Products are not for resale.



What others are saying
There are no contributions yet.