Technote | EGF (Qk011)
Epidermal Growth Factor remains stable and bioactive in conditioned media
Epidermal growth factor (EGF) is an essential growth factor for stimulating the proliferation of induced pluripotent stem cells (iPSC) and embryonic stem cells (ESC) and their subsequent differentiation. EGF is also a key component of many media for developing and maintaining organoids.
In stem cell cultures, the instability of growth factors like EGF contribute to the need for frequent media changes to avoid spontaneous differentiation and variability. The half-life of EGF in conditioned media is believed to be between 1-3 days. Qkine EGF (Qk011) has been tested to ensure its stability in conditioned media.
Qkine EGF (Qk011) stability
- Qkine EGF (Qk011) was bioactive in a quantitative luciferase assay with EC50 of 0.14 ng/ml (22.3 pM).
- After 2-day incubation in conditioned media EGF remained bioactive with an EC50 of 0.10 ng/ml (16.6 pM).
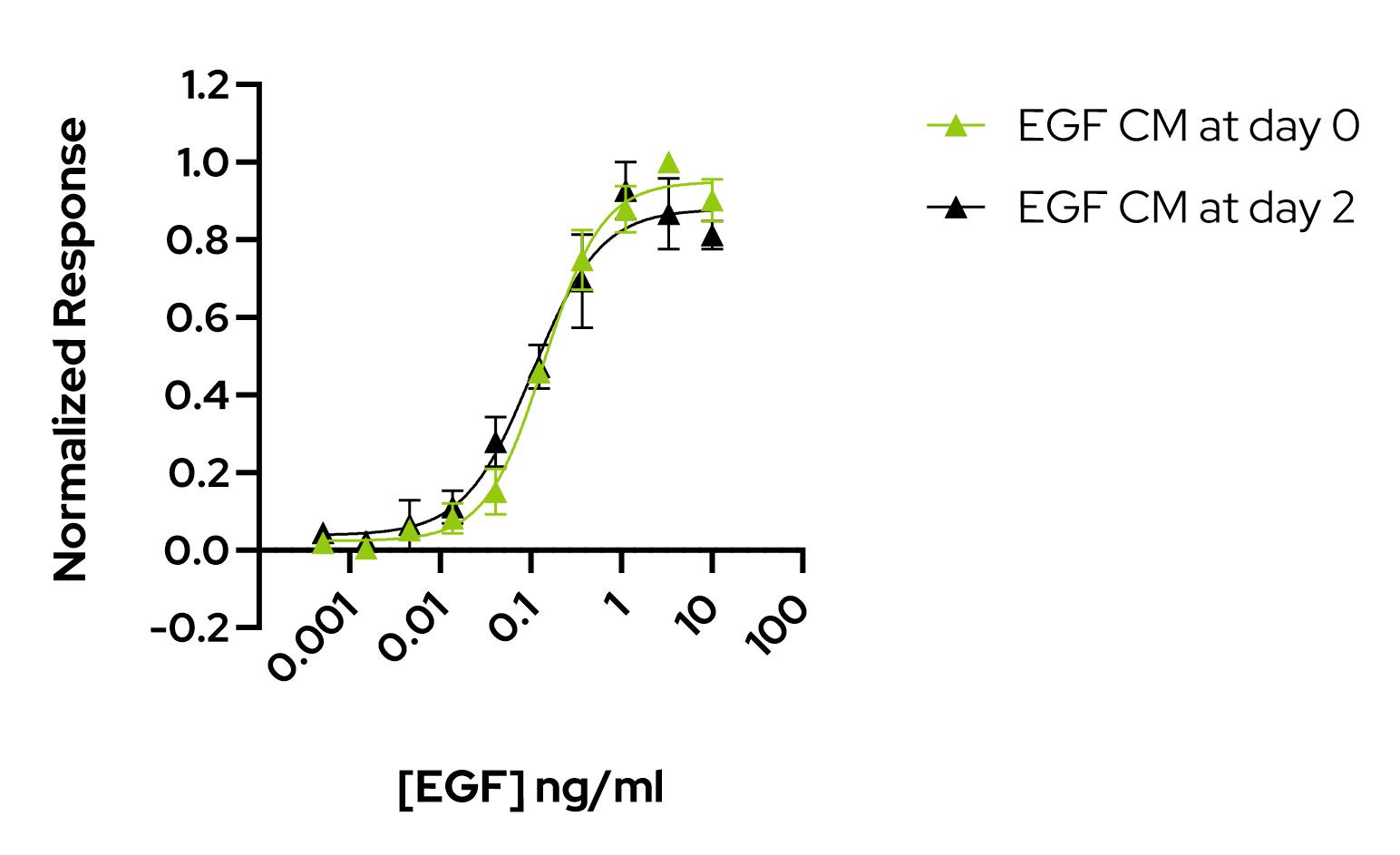
Human EGF remains highly bioactive from day 0 to day 2 in conditioned media. The bioactivity of EGF was determined using the Promega serum response element luciferase reporter assay in HEK293T cells. Cells were treated in triplicate with a serial dilution of EGF for 3 hours after preincubation for 0 days (green) or 2 days (black) in conditioned media.




